ADVERTISEMENTS:
In this article we will discuss about the process of oxygenic and anoxygenic photosynthesis in bacteria.
Oxygenic Photosynthesis in Bacteria:
Light Reaction in Cyanobacteria:
In cyanobacteria (also in all phototrophic eukaryotes), there are two distinct but interconnected photosystems: photosystem I and photosystem II (Fig. 25.4). Photosystem I absorbs longer wavelength light (far-red light) and funnels its energy to a special reaction centre chlorophyll ‘a’ molecule called P700.
The P700 signifies that this reaction centre chlorophyll ‘a’ absorbs light at a wavelength of 700 nm most effectively. Photosystem II absorbs light at shorter wavelengths (near red light) and transfer its energy to the reaction cent-e chlorophyll molecules called P680.
1. Cyclic Photophosphorylation:
When the photosystem I antenna chlorophylls funnel light energy to the reaction centre chlorophyll P700, the latter gets excited and, as a result, its reduction potential becomes very negative. The excited or high-energy electron of P700 is captured by a special chlorophyll ‘a’ molecule (A) or an iron sulphur protein (FeS).
The electron is eventually transferred to ferredoxin. The latter transfers electron to a cyclic route through a series of electron carriers (cytochrome b563 → plastaquinone → cytochrome b6 → cytochrome f → plastocyanin) back to oxidized P700.
Since the electrons travel in a cyclic pathway (i.e. they originate from P700 and come back to the P700), the process is called cyclic photophosphorylation in which only photosystem I is involved. During cyclic phosphorylation, ATP is generated in the region of cytochrome b6.
ADVERTISEMENTS:
2. Non-Cyclic Photophosphorylation.
In this photophosphorylation both photosystem I and II are involved. The reduction potential of P680 chlorophyll a molecule of photosystem II is very electropositive, slightly more positive than that of the H2O/O2 couple. This facilitates the first step in oxygenic electron flow, the splitting of water (photolysis) into oxygen atoms (1/2O2) and hydrogen ions (2H+). Photolysis donates an electron to the oxidized P680 molecule following the absorption of a quantum of light near 680 nm.
The P680 molecule is now excited and reduces pheophytin ‘a’ which is chlorophyll ‘a’ without the magnesium atom. Electrons subsequently travel through quinone, plastaquinone, cytochrome b6 (ATP is generated in the region of cytochrome b6), cytochrome f and plastocyanin; the latter donates electrons to photosystem I.
The electron is accepted by the oxidized reaction centre chlorophyll ‘a’ of photosystem I (P700) which has previously absorbed light quanta and begun the steps to lead the reduction of NADP+ into NADPH.
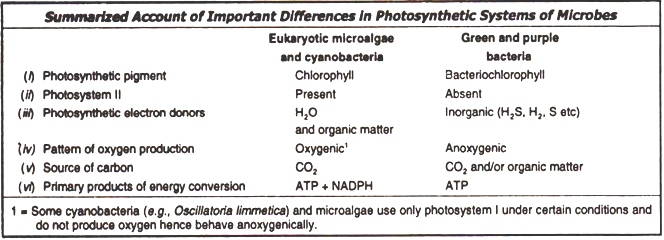
Anoxygenic Photosynthesis in Bacteria:
Purple and green bacteria possess only photosystem I. Since they lack photosystem II, they cannot use water (H2O) as an electron donor in noncyclic photophosphorylation (i.e., noncyclic electron transport) and thus cannot produce oxygen from water photosynthetically, i.e., they are anoxygenic.
Light Reaction in Purple Bacteria:
Light-harvesting antenna bacteriochlorophyll molecules absorb light and transfer it to reaction centre bacteriochlorophyll called P870 (Fig. 25.5). P870 is excited and releases electron which proceeds to reduce a molecule of bacteriopheophytin (Bph) in the reaction centre. This transition completes very fastly taking about three-trillionth of a second (i.e., 3 x 10-12 sec.) time. Once reduced, the bacteriopheophytin reduces several intermediate quinone (Q) molecules to finally, a quinone in “quinone pool”.
This transition is also very fast completing within less than one-billionth of a second. Electrons arc now transported from the quinone through a series of iron-sulphur proteins (FeS) and cytochromes (Cyt) back to the reaction centre (P870).
It is the cytochrome bc1 complex that interacts with the quinone pool during photosynthetic electron flow as a proton motive force (PMF) used to derive ATP synthesis. In addition to ATP, NADP or NADPH are also produced by purple bacteria using H2S (also S2O32-, S0 and even Fe2+) as external electron donors. When H2S is the electron donor, globules of sulphur (S0) are stored inside the cells of purple bacteria.
A reversed electron flow operates in purple bacteria to reduce NAD+ to NADH. The reduced H2S or H2SO32- (thiosulphate) are oxidized by cytochromes and electrons from them eventually end up in quinone pool. However, the energy potential of quinone is insufficiently negative to reduce NAD+ directly. Therefore, the electrons from the quinone pool are forced backward to reduce NAD+ to NADH. This energy requiring process is called reversed electron flow.
Light Reaction in Green Bacteria:
The general scheme of cyclic photophosphorylation is represented in Fig. 25.6. The reaction centre bacteriochlorophyll is P840 that it absorbs light near 840 nm and resides at a significantly more negative reduction potential in comparison to purple bacteria.
Unlike purple bacteria where the first stable electron acceptor molecule resides at about 0.0 reduction potential, the electron acceptors of green bacteria (FeS proteins) reside at about – 0.6 reduction potential and have a much more electronegative reduction potential than NADH.
In green bacteria, ferredoxin reduced by FeS protein serves directly as electron donor for dark reaction (fixation of CO2). Thus, like oxygenic phototrophic microorganisms (and even green plants), in green bacteria both ATP and NADPH are direct products of light reactions.
ADVERTISEMENTS:
When H2S donates electrons to reduce NAD+ to NADH in green bacteria, sulphur globules remain outside of the cell of green bacteria. This is unlike purple bacteria where the globules of sulphur remain inside of the bacterial cell.