ADVERTISEMENTS:
Biotechnology: Various Applications of Biotechnology!
The recombinant DNA technological processes have made great impact in the area of healthcare by mass production of safe and more effective therapeutic drugs.
Further, the recombinant therapeutics does not induce unwanted immunological responses. Now about 30 recombinant therapeutics have been approved for human use all over the world. In India, 12 of these are presently being marketed.
Genetically Engineered Insulin:
Role of Sharpy-Shafer, Banting, Best and Macleod. Sharpy-Shafer (1916) first expressed the opinion that diabetes is caused by failure of the islets of pancreas to secrete a substance named by him as insulin. Insulin is secreted by the Beta cells of the islets of Langerhans of the pancreas. In 1921, Banting and Best succeeded in preparing a pure extract of insulin from the pancreatic islets of a dog with the help of Macleod. Banting and Macleod won the 1923 Nobel Prize in Medicine or Physiology.
ADVERTISEMENTS:
They demonstrated that administration of insulin could cure diabetes in human beings, earlier, insulin for curing diabetes used to be extracted from pancreas of slaughtered pigs and cattle. This insulin is slightly different from human insulin and brings about some undesirable side effects such as allergy.
Structure of Insulin:
Human insulin is made up of 51 amino acids arranged in two polypeptide chains, A having 21 amino acids and В with 30 amino acids. The two polypeptide chains are interconnected by two disulphide bridges (Fig. 12.9) or S-S linkages. An S-S linkage also occurs in A chain. The hormone develops from a storage product called proinsulin.
Proinsulin has three chains, A, В and С. С -chain with 33 amino acids is removed prior to insulin formation. Bacteria cannot be made to synthesise insulin from its gene because of the presence of introns. Bacteria do not possess enzymes for removing intron mediated transcription.
How Insulin is Synthesized?
As stated earlier insulin is produced by the Beta cells of the islets of Langerhans of the pancreas. The gene for this protein synthesis is located on chromosome 11. In mammals, including humans, insulin is synthesized as a pro-hormone (like a proenzyme, the prohormone also needs to be processed before it becomes a fully mature and functional hormone) which contains an extra stretch called the С peptide.
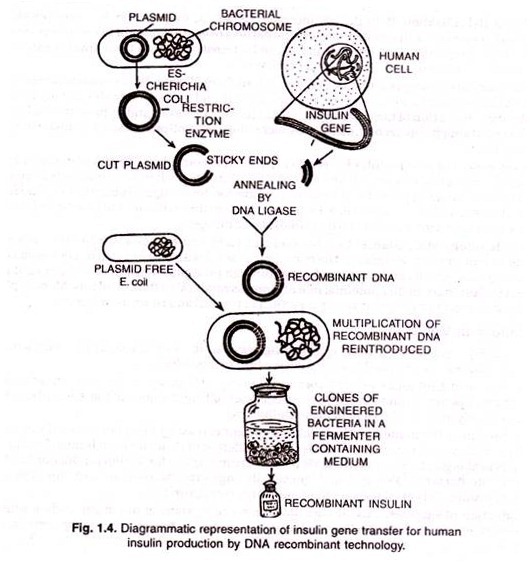
This С peptide is not present in the mature insulin and is removed during maturation into insulin. The main challenge for production of insulin using rDNA technique was getting insulin assembled into a mature form. In 1983, Eli Lilly an American company, first prepared two DNA sequences corresponding to A and В chains of human insulin and introduced them in plasmids of Escherichia coli to produce insulin chains. Chains A and В were produced separately, extracted and combined by creating disulfide bonds to form human insulin (humulin).
Molecular structure of insulin was worked out by Sanger. Tsan synthesised human insulin first time.
Production of Human Insulin:
It involves essentially the following stages:
(i) Isolation of Donor or DNA segment:
A useful DNA segment is isolated from the donor organism.
(ii) Formation of Recombinant DNA (rDNA):
Both the vector and donor DNA segments are cut in the presence of restriction endonuclease. In the presence of ligase DNA segments of both are joined to form rDNA.
(iii) Production of Multiple Copies of rDNA:
ADVERTISEMENTS:
Next step in the process is production of multiple copies of this recombinant DNA.
(iv) Introduction of rDNA in the recipient organism:
This rDNA is inserted into a recipient organism.
(v) Screening of the transformed cells:
ADVERTISEMENTS:
The recipient (host) cells are screened in the presence of rDNA and the product of donor gene.
The transformed cells are separated and multiplied, an economical method for its mass production. The various steps and their sequence for the production of human insulin are depicted in Fig. 12.11.
Dr Saran Narang, a scientist of Indian origin, working in Ottawa, Canada was involved in cloning of insulin gene.
ADVERTISEMENTS:
i. The most famous of the dogs Banting and Best used was one called “Marjorie”.
ii. The first patient of diabetes to be administered insulin was 14- year old Leonhard Thompson.
iii. Insulin cannot be orally administered to diabetic patient because it degrades in the alimentary canal.
iv. Banting was born on November 14th, 1891 hence 14th November is observed as “Diabetic Day”.
ADVERTISEMENTS:
v. In 1982 insulin (Eli Lilly’s Humulin) was the first product made genetically engineered bacteria to be approved for use in Britain and the U.S.A.
Gene Therapy:
What is Gene Therapy?
Gene therapy is the technique of genetic engineering to replace ‘a faulty gene’ by a normal healthy functional gene.
Types of Gene Therapy:
It is of two types:
(i) Germline Gene Therapy:
ADVERTISEMENTS:
In this type of gene therapy germ cells, i.e., sperms or eggs (even zygotes) are modified by the introduction of functional genes, which are ordinarily integrated into their genomes.
(ii) Somatic Cell Gene Therapy:
In this type of gene therapy, the gene is introduced only in somatic cells.
Only introduction of a new gene into the somatic cells is allowed at present. Genetic modification in the germ cells of the offspring is not permissible.
Diseases and Gene Therapy:
The diseases for which scientists are making serious attempts to control through gene therapy are severe combined immunodeficiency (SCID) disease, Duchenne muscular dystrophy and cystic fibrosis. These disorders are mainly due to single gene defects. Cancer, cardiovascular diseases, diabetes, hypertension, arthritis, sickle cell anaemia, etc. are complex genetic disorders. However, the day is not far off when these diseases can be cured through gene therapy.
Example. Adenosine Deaminase (ADA) Deficiency:
ADVERTISEMENTS:
The first clinical gene therapy was given in 1990 to a 4-year old girl with adenosine deaminase (ADA) deficiency. This enzyme is very important for the immune system to function. ADA deficiency can lead to severe combined immune deficiency (SCID). SCID is caused due to defect in the gene for the enzyme adenosine deaminase.
In some children ADA deficiency can be cured by bone marrow transplantation. However, in others it can be treated by enzyme replacement therapy, in which functional ADA is given to the patient by injection. But in both approaches the patients are not completely cured.
Because these patients do not have functional T-lymphocytes, they cannot provide immune responses against invading pathogens.
As a first step towards gene therapy (Fig. 12.12), lymphocytes, a kind of white blood cells, are extracted from the bone marrow of the patient and are grown in a culture outside the body. A functional ADA cDNA (using a retroviral vector) is then introduced into these lymphocytes, which are re injected to the patient’s bone marrow.
But as these cells do not always remain alive, the patient requires periodic infusion of such genetically engineered lymphocytes. However, if the isolated gene from bone marrow cells producing ADA is introduced into cells at early embryonic stages, it can be a permanent cure.
Molecular Diagnosis (Diagnosis of Disease):
It is well known that an early diagnosis and understanding its pathophysiology (symptoms, etc.) are very important for the effective treatment of the disease. Using conventional methods of diagnosis (serum and urine analysis, etc.) early detection is not possible. Recombinant DNA technology, Polymerase Chain Reaction (PCR) and Enzyme Linked Immunosorbent Assay (ELISA) are some of the techniques that serve the purpose of early diagnosis.
The molecular probes are usually single stranded pieces of DNAs (sometimes RNAs) labelled with radio isotopes such as 32p. Molecular probes are available for many genetic disorders such as Duchenne muscular dystrophy cystic fibrosis, Tay-Sachs disease.
The analytical techniques used for the identification of a specific DNA, an RNA or a protein from thousands of each are collectively called blotting technique. In Southern blotting extraction of DNA from the cells (say leucocytes) occurs. Latter on labelled DNA hybrid complexes are formed which can be identified on exposure to X-ray film.
In Northern blotting RNA is identified by labelled DNA or RNA probe. In Western blotting, protein is identified with the help of labelled antibody probe. The radioactively labelled DNA probes are formed.
Presence of a pathogen (bacteria, viruses, etc.) is usually suspected only when the pathogen has produced a diseased symptom. By this time the number of pathogens is already very high in the body, but very low count of a bacteria or virus (when the symptoms of the disease are not yet visible) can be detected by multiplication of their nucleic acid by PCR.
PCR can detect very low amounts of DNA. PCR is now usually used to detect HIV in suspected AIDS patients. It is also used to detect mutations in genes in suspected cancer patients. It is a good technique to identify many other genetic disorders.
A single stranded DNA or RNA joined with a radioactive molecule (probe) is allowed to hybridize to its complementary DNA in a clone of cells. It is followed by detection using autoradiography. The clone having the mutated gene will not appear on the photographic film, because the probe will not have the complementarily with the mutated gene.
ELISA is based on the principle of antigen-antibody interaction. It can detect very small amount of protein (antibody or antigen) with the help of enzyme (e.g., peroxidase or alkaline phosphatase). Infection by pathogen can be detected by the presence of antigens such as proteins, glycoproteins, etc. or by detecting the antibodies synthesised against the pathogen.
Steriods:
Steroids are crystallisable lipids of high molecular weight. They consist of one 8-carbon ring and three 6-carbon rings. Steriods are found in both plants and animals. Murray and Peterson (1950) observed that the fungus Rhizopus stolonifer could bring about hydroxylation of steroids. Important uses of steroids are:
(i) Cortisone and its derivatives (prednisone and prednisolone) are effective in the treatment of rheumatoid arthritis.
(ii) Steroid hormones are known to be regulators of metabolism in the animal or human body.
(iii) Steroid treatment is given to suppress immune responses in patients with autoimmune diseases or persons who have had organ transplants.
(iv) Prednisolone is used as an anti-inflammatory drug.
(v) Oestrogens and progesterone’s are employed in the preparation of oral contraceptives (birth-control pills).
Vaccines:
Vaccines are either attenuated (live but weak) or dead (inert) agents of disease which when administered into a healthy person provide temporary or permanent immunity to that particular disease. The term vaccine was introduced by Edward Jenner (1790) who worked on small pox.
Later, Jenner’s findings were extended by Louis Pasteur (1879) to other infective diseases such as anthrax, rabies and cholera. Pasteur established the scientific basis of vaccination. Recently some second generation vaccines have been prepared with the help of genetic engineering technique against hepatitis-B and herpes virus.
They are more uniform in quality and produce less side effects as compared to ‘first-generation the vaccines. Now-a-days production of ‘third generation vaccines’ called synthetic vaccines, are being tried. Antifertility vaccines have also been developed.
The genes encoding antigenic proteins can be isolated from the pathogens and expressed in plants. Such transgenic plants or their tissues producing antigens can be eaten for vaccination/immunization. These are called edible vaccines.
The expression of such antigenic proteins in crops like banana and tomato are useful for immunization of humans because banana and tomato fruits can be eaten raw. The edible vaccines that are produced in transgenic plants have great advantages like less storage problems, easy delivery system by feeding and low cost as compared to the recombinant vaccine.
Human Growth Hormone (hGH)—Somatotropin:
hGH is secreted by the anterior lobe of pituitary gland. Secretion of hGH is regulated by two other hormones secreted by hypothalamus. These hormones are:
(i) somatotropin releasing hormone which stimulates the anterior lobe of pituitary gland to release somatotropin or growth hormone
(ii) Somatostatin or growth inhibiting hormone which inhibits the secretion of growth hormone from the anterior lobe of pituitary gland. Deficiency of somatotropin in about 3% cases is hereditary. It has been estimated to about 1 child in 5,000. hGH is very useful to the children born with hypopituitarism which is a form of dwarfism. It is caused by less secretion of hGH from the anterior lobe of pituitary gland. hGH is also helpful in “healing of injuries”.
Bovine Growth Hormone (BGH):
This hormone has veterinary uses. For example, injecting a cow with bovine growth hormone (BGH) can increase milk production by as much as 25%. BGH also improves beef yield in cattle (10-15% increase in body mass).
Forensic Medicine (Identification of Murders, Rapists, etc.):
Biotechnology has proved to be a boon in solving crimes, legal disputes, etc. Establishing the identity of victims (e.g., of murder, accidents, etc.), criminals (e.g., in cases of rape, murder, etc.), father (in cases of paternity dispute) etc. is unable to solving the problems of crimes/cases. A biotechnological procedure, called DNA finger-printing or DNA profiling, is a highly sensitive, fool-proof, absolutely accurate and extremely versatile approach to this problem.
Monoclonal Antibodies:
Antibodies derived from a single clone of cells which recognize only one kind of antigen, are called monoclonal antibodies. The technique of producing monoclonal antibodies by Fusing normal antibody-producing cells with cells from cancerous tumors was introduced by Georges Kohler and Cesar Milstein in 1970. The major steps in the production of monoclonal antibodies with hybrid cultures are mentioned below:
(i) First of all a mouse rat or some other animal is injected with specific antigen (against which the antibodies are required).
(ii) The animal starts developing antibodies against the antigen in B-lymphocyte cells in spleen,
(iii) The spleen of animal is removed and its B-lymphocyte cells are isolated
(iv) Similarly, the cells producing bone marrow cancer (myeloma cells) are isolated. These cells should not be able to synthesise their own nutrients,
(v) The two types of cells (i e myeloma cells and antibody-producing cells) are made to fuse in cultures. The fused cells are called hybridomas.
(vi) The entire culture is shifted to a medium deficient in the nutrient needed by the myeloma cells where myeloma cells cannot survive. In this medium all the un-fused myeloma cells die and only hybridoma cells survive, (vii) The surviving hybridoma cells are allowed to multiply separately and each clone is tested for its ability to produce a desired antibody.
(viii) The clones which show positive results are isolated and cultured for large scale production of the antibody.
Monoclonal antibodies are highly specific for specific antigens and can be easily cultured outside the body. These antibodies are, therefore, more effective and ideal for diagnosis of some specific diseases. One of the most effective applications of monoclonal antibodies is immune suppression for kidney transplantation.
Interferons (IFNs):
Interferons are the antiviral glycoproteins (called cytokines) functioning as immune regulators or lymphokines produced by the infected cells in response to viral infections (discovered in 1957 by Alec Issacs and Jean Lindenmann). These proteins are produced by most body cells on exposure to viruses.
They diffuse to neighbouring cells and trigger a reaction that neutralizes the particular viruses. Some interferons also neutralize other viruses and, therefore, prevent viral infections. They also inhibit cellular proliferation and modulate the immune system of the organism.
There are three major classes of interferons
(i) Interferons-a (IFN-a):
This kind of interferon is produced when leucocytes and lymphocytes are exposed to virus.
(ii) Interferon-b (IFN-b):
These are produced by fibroblasts, epithelial cells, macro- phytes and leucocytes in response to viral infection.
(iii) Interferon-g (IFN-g):
These are produced by T-lymphocytes induced by antigenic stimulation.
Until recently the only source of interferons was human white blood cells or virus infected human cells grown in tissue culture. Production of human interferon by cloning of genes in colon bacilli was started in 1980 by two American scientists Gilbert and Weissmann.
The work triggered a wave of experimentation resulting in the production of interferons in large amounts by recombinant DNA technology. The interferons (particularly IFN-a) are used on a significant scale for the treatment of hepatitis-B. Interferons are also being tested for the treatment of cancer and some viral diseases including AIDS.
Use of Polymerase Chain Reaction (PCR):
PCR is a technique by which any piece of DNA can be quickly amplified (copied many times) without using cells. The DNA is incubated in a test tube with a special kind of DNA polymerase enzyme, a mixture of deoxyribonuleotides for use as raw material, and short pieces of synthetic single stranded DNA to serve as primers for DNA synthesis. PCR can make billions of copies of a DNA segment in a few hours.
To clone a DNA segment in bacteria takes days. When the source of DNA is scanty or impure, it can be amplified by PCR technique. Amplification can make the identification of DNA easier. Since the sequence of HIV DNA is known, its amplification by PCR can help detect HIV DNA in blood or tissue samples.
This is often the best way to detect infection. DNA from single embryonic cell is amplified by PCR for rapid prenatal diagnosis of genetic disorders. DNA from tiny amounts of blood, tissue or semen found at the site of crime can be amplified to facilitate its identification.
DNA technology can help identify individuals with genetic disorders before the appearance of symptoms, even before birth. It is also possible to identify symptomless carriers of potentially harmful recessive alleles, such as of haemophilia, phenylketonuria (PKU). PCR is also helpful in DNA fingerprinting.
Making a Choice of Baby’s Sex:
Recently techniques have also been developed which will not require preferential abortion but will allow preferential fertilization by male (carrying Y chromosome) or female (carrying X chromosome) determining sperms. There are techniques available now, which allow separation of sperms carrying Y chromosomes, from the ejaculate of a man (through Ericson’s method developed by R. Ericson of U.S.A) to be used for insemination of ovulating women.
This technique (using quinacrine stain) has been used with 80% success in 47 sperm centres in the world including one in Mumbai. Ericson has actually established a company named Gametrics Ltd, in California, U.S.A. which specializes in separating sperms with Y chromosome and hundreds of male children have been produced with its help.
Techniques have also been developed to separate sperms carrying X chromosome for artificial insemination leading to the birth of female children. This technique involves ‘sephadex gel column’ in which sperms with Y, being lighter are trapped in gel and those with X being heavier reach the bottom of the column, and can be used for inseminaton.
Production of Vitamins:
1. Vitamin B12 (Riboflavin):
Riboflavin is produced commercially by direct fermentation utilizing the fungus Ashbya gossypii. Fermentation conditions -pH 6.0 to 7.5, 4 to 5 days at 28-30°C, aerobic.
2. Vitamin B12 (Cobalamine):
Now a day’s vitamin B12 is produced by a direct fermentation utilizing streptomyces species such as Streptomyces griseus. Fermentation conditions -pH 6 to 7.7, 2 days at 26 – 28°C, aerobic.
3. Vitamin С (Ascorbic Acid):
Vitamin С was first recognized when in 1747, Scottish naval surgeon James Lind discovered that something in Citrus foods prevented scurvy. In 1928 Albert Szent-Gyorgyi, a much admired biochemist, was the first to isolate vitamin С (ascorbic acid). He later won the 1937 Nobel Prize in Physiology and Medicine for other work.
Vitamin С was the first vitamin to be synthesized artificially in a process invented by Dr. Tadeusz Reichstein, of the Swiss Institute of Technology in Zurich in 1939. Vitamin С is produced by utilizing Gluconobacter oxydans. Fermentation conditions -pH 7, 45 hours at 30°C, aerobic.
4. Vitamin A (Р-Carotene):
P-carotene is produced by members of choanephoraceae family of phycomycetes. Phycomyces blakesleeanus, Choanephora cucurbitarum and Blackeslea trispora have been extensively studied for their ability to produce P-carotene.
Applications of Recombinant DNA Technology/Genetic:
1. Molecular Analysis of Diseases:
DNA research has helped in understanding the molecular basis of diseases like sickle cell anaemia, thalassemias, etc.
2. Production of Proteins in Abundance:
Using recombinant DNA technique several proteins have been produced in abundance for curing the diseases. These are insulin, growth hormone, interferon’s, vaccines, erythroprotein and blood clotting factors.
3. Laboratory Diagnostic Application:
rDNA technology makes the diagnosis of many diseases (e.g., AIDS) simple and quick.
4. Gene Therapy:
The genetic diseases like sickle cell anaemia can be cured through gene therapy.
5. Prenatal Diagnosis of Diseases:
DNA collected from the amniotic fluid surrounding the foetus can be used for predicting the genetic diseases.
6. Application to Forensic Medicine:
rDNA technology has greatly helped to identify criminals by DNA fingerprinting and settle the disputes of parenthood of children.
7. Agricultural Application:
rDNA technology is used for developing transgenic plants which resist drought and diseases and increase their productivity. It improves quality of food.
8. Industrial Application:
Enzymes synthesized by rDNA technology are used to produce sugars, cheese and detergents.
9. Application to Animals:
It is used for developing test tube babies to overcome infertility and production of transgenic animals.
10. Evolution:
rDNA technique is of great use in joining several missing links in the evolution. This is done by amplifying the DNA of extinct animals.
Ethical Issues:
Ethics includes rules of conduct by which a community regulates its behaviour and decides as to which activity is lawful and which is not. Therefore, bioethics includes rules of conduct that may be used to regulate our activities in relation to the biological world.
The main bioethical concerns pertaining to biotechnology are briefly mentioned as follows:
(i) Introduction of a transgene from one species into another species violates the ‘integrity of species’,
(ii) Biotechnology may pose unforeseen risks to the environment, including risk to biodiversity,
(iii) Transfer of human genes into animals (and vice-versa) dilutes the concept of ‘humanness’,
(iv) When animals are used for production of pharmaceutical proteins, they are virtually reduced to the status of a ‘factory’,
(v) Use of animals in biotechnology causes great suffering to them,
(vi) Biotechnology is disrespectful to living beings, and only exploits them for the benefit of human beings.
Therefore, the Indian Government has set up organisations such as GEAC (Genetic Engineering Approval Committee), which will make decisions regarding the validity of GM research and the safety of introducing GM-organisms for public services.
1. Certain companies are being granted patents for products and technologies that make use of the genetic materials, plants and other biological resources that have long been identified, developed and used by farmers and common persons of a particular region/ country. There are numerous varieties of rice in India alone.
The diversity of rice in India is one of the richest in the world. Basmati rice is distinct for its unique aroma and flavour and 27 documented varieties of Basmati are grown in India. There is reference to Basmati in ancient books, as it has been grown for centuries. In 1997, an American company got patent rights on Basmati rice through the US Patent and Trademark Office.
This allowed the company to sell a ‘new’ variety of Basmati, in the US and abroad. This ‘new’ variety of Basmati had actually been derived from Indian farmer’s varieties. Indian Basmati was crossed with semi-dwarf varieties and claimed as an invention or a novelty. The patent extends to functional equivalents, implying that other people selling Basmati rice could be restricted by the patent.
2. Several attempts have also been made to patent uses, products and processes based on Indian traditional herbal medicines, e.g., turmeric neem. If we are not vigilant other countries/individuals may encash on our rich legacy.
Mostly the developed nations are rich financially but poor in biodiversity and traditional knowledge. In contrast the developing and the underdeveloped world is rich in biodiversity and traditional knowledge related to bio-resources. Traditional knowledge related to bioresources can be exploited to develop modern applications.
Some nations are developing laws to prevent such unauthorised exploitation of their bioresources and traditional knowledge.
3. The GM crops are fast becoming a part of agriculture throughout the world because of their contribution to the increased crop productivity and to global food, feed and fiber security, besides their use in health-care and industry.
4. The effect of GM crops on non-target and beneficial insects/microbes.
5. Transgenes may escape through pollen to related plant species (gene pollution) and may lead to the development of super weeds.
6. The GM crops may change the fundamental vegetable nature of plants as the genes from animals (e.g., fish or mouse) are being introduced into crop plants.
7. The safety of GM food for human and animal consumption, (e.g., GM food may cause allergenicity).
8. The effect of GM crops on biodiversity and environment.
9. The transgenes may move from plants to gut microflora of humans and animals and cause antibiotic resistance.
10. The GM crops may lead to the change in the evolutionary pattern.
11. Scientists cannot rule out the possibility of mutation or other biological damage.
12. The release of genetically engineered plants and animals in the environment could disturb the existing ecological balance.
13. The use of recombinant microorganisms for various commercial purposes can accidentally create new infectious agents.
14. The main fear associated with the genetically engineered microorganisms is that they could escape from the laboratory into the environment with unpredictable fatal consequences. AIDS virus is supposed to be the outcome of such a research.
Bio-patent:
A patent is the right granted by a government to an inventor to prevent others from commercial use of his invention. When patents are granted for biological entities and for products derived from them, these patents are called biopatents. Primarily, industrialised countries, like U.S.A., Japan and members of European Union, are awarding Bio-patents. Bio-patents are awarded for (i) strains of microorganisms, (ii) cell lines, (iii) genetically modified strains of plants and animals, (iv) DNA sequences, (v) the proteins encoded by DNA sequences, (vi) various biotechnological procedures, (vii) production processes, (viii) products and (ix) product applications. There is an opposition from social groups to biopatents. These objections are mainly ethical and political. Some biopatents are very broad in their coverage. For example, one patent covers “all transgenic plants of Brassica family”.
Cancelled patents on natural product inventions:
Patents on natural product inventions are subject to attack unless all public knowledge about the species in question and its use are fully disclosed. For example, a 1995 patent, “Use of Turmeric in Wound Healing”, was cancelled in 1998. The new evidence established that use of turmeric to promote wound healing had been known for generations in India.
Likewise, the 1986 plant claimed an ostensibly new, distinct variety of Banisteriopsis caapi, known in the Amazon as ayahuasca. However, new evidence establishes that the claimed plant is actually the wild uncultivated type, and is neither new nor distinctive. COICA, an organization of indigenous people, and the Amazon Coalition have requested re-examination of the ayahuasca patent, seeking to eliminate what is perceived as an immoral expropriation of their traditional and biological heritage. More such challenges can be anticipated.
Significance of Bio-patents:
Bio-patent system allows private, monopoly rights over cells, genes, animals and plants. It means that people will not share vital research information because they are afraid that it will be patented by someone else. The people will not research in areas that are dominated by patents.
It will lead to research programmes dominated by patentability and profitability rather than need. It gives the patent holder monopoly control over resources for food and medicine. The important advantages of bio-patents are that they are a direct incentive for genetic engineering. The arguments in favour of bio-patents are primarily increased economic growth.
Genes, cells, microorganisms, plants and animals are not an invention and, therefore, should not be patented.
Bio-piracy:
Some organisations and multinational companies exploit and/or patent biological resources or bio-resources of other nations without proper authorisation from the countries concerned, this is called bio-piracy.
Historical Aspects:
Some examples of famous collecting trips are given below:
(a) The recorded history of international plant collecting missions goes back at least 3500 years when Egyptian rulers began bringing plants home after military expeditions.
(b) In the last century, the British Empire instituted regular plant collections. During the Voyage of the Beagle, Charles Darwin simply took what interested him, from the Galapagos and elsewhere, and brought it home.
(c) The Royal Botanical Gardens took rubber trees from Brazil, and planted them in South East Asia. They took cinchona seeds from Bolivia, in violation of national law, and planted them in India.
(d) Commodore Perry’s naval mission to Japan collected a wide variety of plants to bring back to the United States.
(e) More recently, the adventures of Richard Shultes during the mid-twentieth century have become a legend among ethnobotanists. He was able to befriend local shamans, who allowed him to collect thousands of voucher specimens of medicinal plants, hundreds of which had never previously been identified taxonomically.
None of these famous collecting trips was challenged on legal grounds. If done today, how would they be challenged?
Exploitation of Bio-resources:
Institutions and companies of industrialised nations are collecting and exploiting the bio-resources, as follows:
(i) They are collecting and patenting the genetic resources themselves. For example, a patent granted in U.S.A. covers the entire ‘basmati’ rice germplasm indigenous to our country,
(ii) The bio-resources are being analysed for identification of valuable bio-molecules. A bio-molecule is a compound produced by a living organism. The bio-molecules are then patented and used for commercial activities,
(iii) Useful genes are isolated from the bio-resources and patented. These genes are then used to generate commercial products,
(iv) The traditional knowledge related to bio-resources is utilised to achieve the above objectives. In some cases, the traditional knowledge itself may be the subject of a patent.
Brazzein:
Brazzein is a protein produced by a West African plant, Pentadiplandra brazzeana which is approximately 2,000 times as sweet as sugar. It is used as a low calorie sweetener. Local people have been using the super-sweet berries of this plant for centuries. But the protein brazzein was patented in U.S.A. The gene encoding brazzein was also isolated sequenced and patented in U.S.A. It is proposed to transfer the brazzein gene into maize and express it in maize Kernels. These Kernels will then be used for the extraction of brazzein. This development could have serious implications for countries exporting large quantities of sugar.
Examples of Bio-piracy:
The Thai Ministry of Science and its Biotech Institute has accused the British University of Portsmouth of “bio-piracy” as they have refused to return up to 200 strains of marine fungi that they collected in coastal waters and swamps around Thailand. Instead, Portsmouth University is reported to be in the process of selling the rights on “their” Thai fungi to a drug company for screening, as the fungi are believed to contain compounds for treating everything from AIDS to cancer- worth millions of pounds. Thailand insists that keeping the fungi without permission is in breach of international agreements.
India is a country rich in tradition, communal knowledge and expertise in natural medicines, spices, food preparations, biological pesticides and diverse agriculture. It is thus under siege from bio-pirates. Through patenting without consent, foreign companies have collared at least 22 plants for their beneficial derivatives. Patents have been taken out on plants such as black pepper (Piper nigrum), basmati rice (Oryza sativa), Indian mustard (Brassica campestris), pomegranate (Punica granataum), turmeric and neem. US, Japanese and German companies are the principal patenting pirates.
Nestle India has lined up a processing patent on cooked cereals, vegetable pulao and parboiled rice, at the Indian Patent Office, even though Indians have been making parboiled rice, often as a staple diet for centuries.
Bio-war:
War which is fought by bioweapons (biological weapons) against humans, their crops and animals is called biowar (biological war). Viruses, bacteria, fungi, spores and toxins can be used as bio-weapons (BW). Bio-weapons used during the 20th century are (i) 1942 Bntain developed strategic amounts of anthrax, (ii) 1940s Nazi prisoners infected with Rickettsia spp. hepatitis A, plasmodia spp. and treated with investigational vaccines and drugs, (iii) 1940s Nazi secret agent Reinhardt Heydrich assassinated with botulinum toxin, (iv) Germany used Bacillus anthracis (anthrax) to infect livestock and animal feed exported to Allies, (v) 1932-1945 Japan conducted research on B. anthracis, Shigella spp. V. cholera and Y. pestis.
US Bio-weapons Program can be mentioned as follows (i) In 1942 approximately 5000 bombs filled with B. anthracis produced, (ii) In 1950s program expanded during Korean War.
(iii) In 1955 human experiments with F. tularensis and C. burnetti. (iv) In 1949-1968 simulant organism released off coast of San Francisco and in New York City subway, (v) In 1969 US terminated offensive bio-weapons program, (vi) In 1972 Bio-weapons Convention and Treaty.
Diseases caused by bio-weapon agents are (i) Anthrax (ii) Small pox (iii) Plague (iv) Q- fever (fever, chills fatique, etc.) (v) Tularemia (it can occur in humans in two forms: ulceroglandular and typhoidal). (vi) Viral Encephalitis, (vii) Viral haemorrhagic fevers, (viii) Botulinum toxin (skeletal muscle paralysis).
Why are bio-weapons used? (i) Bio-weapons are low cost weapons, (ii) They cause far more casualties than chemical or conventional weapons. Once bio-weapon agents are released they are invisible, odourless and tasteless.
Protection against bio-weapons, (i) Use of respirator or gas mask, (ii) Protective shelter, (iii) Decontamination, (iv) Vaccination, (v) Antibiotics.
Biosafety Issues:
Measures taken to prevent any risk to plants, animals and microbes from transgenic organisms are known as biosafety. It was feared that genetically engineered microorganisms (GEMS) may disturb the ecosystem and its processes, in which they might be released. They may rapidly multiply and outcompete the native microbes. They may also transfer genes related to virulence or pathogenesis into bacterial population and, thereby increase their virulence. Similarly, genetically modified plants could pose biological and ecological risk.
Discussions on possible hazard of cloning recombinant DNA molecules began in the early 1970s. The main concerns were examined by a committee of National Academy of Sciences (USA) in 1974. The National Institute of Health (NIH), USA established the Recombinant Advisory Committee (RAC) in 1974. In Febmary 1975, a historic international meeting was convened at Asilomar, California.
The first NIH guidelines were prepared in 1975, they were more strict than the recommendations of the Asilomar Conference by 1981; most cloning experiments in E. coli, K-12, certain strains of Bacillus subtilis and Saccharomyces cerevisiae were considered exempt from other requirements of NIH guidelines. A major revision of the guidelines was done in 1982; containment levels were lowered and experiments that were previously prohibited were changed and approved by NIH.