ADVERTISEMENTS:
This article throws light on the six important fungal products obtained from fungal biotechnology.
The six products are: (a) Food and Beverages, (b) Fungal Secondary Metabolites, (c) Fungal Enzymes, (d) Biocontrol Agents and Other Uses, (e) Application of Molecular Biology in Fungal Biotechnology, and (6) Future Prospects.
A. Food and Beverages:
(i) Production of Alcoholic Beverages:
Under anaerobic or oxygen scarce situation and if the sugar concentration is high, yeast ferment to produce ethanol and CO2. This alcoholic fermentation is the basis of the production of the great variety and huge quantities of alcoholic beverages consumed. The yeast responsible is Saccharomyces cerevisiae.
ADVERTISEMENTS:
On the Basis of Producing Materials Alcoholic Beverages are of three types:
(i) Wine and cider,
(ii) Beer, and
(iii) Spirits and fortified wines.
ADVERTISEMENTS:
Wine:
The major wine producing countries have laws that define wine as the product of the alcoholic fermentation by S. cerevisiae of the fresh grape juices. There is, of course, one exception,—Botrytis cinera, a plant pathogen, during their fermentation first send their hyphae into the skin of the grapes causing water loss, resulting in shriveled grapes with unusually high sugar concentration.
This is next allowed to ferment by S. cerevisiae and the so—called French Pourifure noble or the German Edelfaule is formed. The differences between the wines depend on the variety of the grape, the local soil and climate, the properties of the yeasts and the details of the production method.
The fermentation is traditionally carried out in open vats but fermenters giving a greater control of conditions are now widely used, especially for white wines. The yeast primarily responsible for alcoholic fermentation is Saccharomyces cerevisiae. The sugar rich grape juice and other nutrients permit rapid yeast growth which soon results in anaerobic conditions. Ethanol is produced more and more during fermentation until alcohol concentration of 10-12% (v/v) is reached.
Depending on how much sugars remains unutilized, a sweet, medium or dry wine results. After the first phase of fermentation, wine is transferred to wooden casks for maturation and storage. Microbiological activity continues at a reduced level during maturation to form blander lactic acid for rising of pH. This reaction is very desirable where the grape juice is strongly acidic.
The production of cider from apple juices and perry from the juices of pears follow the same techniques and organism for fermentation. In some tropical countries like India, palm wines, made from the sap of a variety of palms, are of considerable nutritional and social importance. The Mexican equivalent of palm wine pulque, is also very important.
Beer:
Beer is basically an alcoholic beverage made from malted barley and flavoured with hops (female flowers of Humulus lupulus). Beers are of two main types—ale and lager. Ale is produced by the top-yeast, served at room temperature and is restricted to British Isles. On the. other hand, lager is produced by the bottom yeast, served well- chilled throughout the world.
Before fermentation malting is done by adding warm water to the coarse flour of malt. This initiates barley enzymes to break the starch and proteins in the malt. After few hours it was boiled to stop enzymatic activities and the hop is mixed to add flavour.
ADVERTISEMENTS:
In traditional fermenters the yeasts mixed with gas accumulates as a foam at the top of the vat during ale production. Lager is produced with a bottom yeast which remained at the bottom as a result of their more hydrophilic cell walls which tends them to sediment. In the brewing industry the lager yeast is termed S. carlsbergensis and the ale yeast is termed S. cerevisiae. After maturation, fining is done to clarify the beer to get the final product.
Spirits:
A wide range of spirits are made from sugary or starchy material. Among the former is brandy (from grape juice), fruit brandies such as applejack (apple juice), rum (juice from sugarcane), tequila (juice from agave) and a range of spirits corresponding to the various palm wines. Among those from starchy materials are whisky from Scotland and whiskey from elsewhere.
A variety of cereals are used for their manufacture, including barley, wheat, rye and maize. Gin is also made from rye and vodka is made from potatoes. The most important organism responsible for fermentation is S. cerevisiae.
ADVERTISEMENTS:
The microbes like Schizosaccharomyces and Clostridium have an important role in flavour formation. Distillation can be carried out with pot stills. Distillation of a ‘wine’ or ‘beer’ with an ethanol concentration of 8-12% can give a condensate with an ethanol concentration of 25-32%. Higher ethanol concentration can be obtained by distilling the condensate.
(ii) The Cultivation of Fungi for Food:
Fungi may be used to modify food to make it more nutritious. They may also be used to produce materials of nutrition value such as vitamins, amino acids and lipids. In addition, the macro fungi may be cultivated to yield fruit bodies (mushrooms), and yeast cells and mold mycelium grown in fermenters for food (single cell Protein).
Mushrooms:
The cultivation of the common white mushroom Agaricus bisporus has expanded worldwide and accounts for over 70% of all mushrooms produced and consumed. The USA continues to be the world’s largest producer. However, mushrooms traditionally grown in the Far East—such as Lentinula edodes (the Shiitake mushroom), Pleurotus sp. (the oyster mushroom), and Volvariella volvaceae (the padi-straw mushroom)—are now expanding into other areas of the world largely because of their unique flavours and textures and amenability to culinary inclusion.
ADVERTISEMENTS:
Mushroom production is, in principle, a fermentation process. In the case of Agaricus the substrate for growth is straw while for Lentinula it is wood. For Agaricus cultivation the straw is composted with animal manures and other organic nitrogen compounds over a period of one to two weeks and the final product is a unique substrate suitable for the rapid growth of the Agaricus inoculum.
There is no standard pattern for compost, formulation being based only on the availability and price of the raw materials and supplements in the particular growing region. The nature of the substrate and its pretreatment, more than all other aspects of growing, determines the method by which specific mushrooms are cultivated.
When the mushroom mycelium (thread-like biomass) has grown throughout the prepared compost (usually contained in large wooden boxes), the environmental conditions of temperature and humidity are altered and subsequently the large mushroom structure forms rapidly in large numbers or ‘flushes’.
These are then harvested by hand cutting and approximately 7-10 days later another crop will appear. Usually, up to four crops are produced before the process is terminated. The cultivations of Volvariella and Pleurotus are straw based but have a much more simple procedure.
ADVERTISEMENTS:
Lentinula edodes is the second-most cultivated mushroom in the world and has been farmed for over 2,000 years. Currently, over 90% of its production occurs in Japan but cultivation extends to China, Korea, Singapore, Taiwan, Sri Lanka and, more recently, into the USA and Europe.
The principal method of cultivation has been to inoculate wooden logs (2m ∞ 15cm) with spore inoculum or mycelial plugs, allow the logs to stand for up to nine months to achieve colonization by the fungus and then, during subsequent early summer and autumn periods, the mushrooms will grow out and be harvested.
This seasonal production is augmented by drying mushrooms to achieve all-year-round consumption. The fungus derives its total nutrition from the lignocellulose of the log. More recently, a new method of cultivation has been developed in which sawdust is mixed with cereal supplements and compressed into large plastic bags — artificial log production.
The bags are sterilized and then inoculated with the pure fungus and, after a period of vegetative growth, are induced to produce the mushroom. This controlled form of cultivation is leading to a wider geographical range of commercial cultivation—not only for Lentinula but for many others.
Undoubtedly the great advantage of these mushroom fermentations is the possibility of converting industrial, urban and wood wastes into a product that is directly edible by humans. However, irrespective of public demand and relative nutritional values, the future outlook for these processes will be related largely to the economics of production methods.
Protoplast Technology in Mushroom:
ADVERTISEMENTS:
Improvement of mushroom cultivation is possible by accumulating desired phenotypes together. Unlike yeasts, gene transfer in filamentous fungi as well as in mushrooms is not so simple and the rate of success is very less. The discovery of technology based on isolated protoplasts gave a new impetus to the improvement of strains of mushroom of commercial importance. Gene transfer using protoplasts has been developed to break down the barrier of gene exchange imposed by conventional breeding system.
In protoplast technology the fungal cell walls are first digested away by the enzymes of microbial origin which leads to form isolated naked protoplasts. Isolated protoplasts are very much susceptible to osmotic stress.
So, these are made osmotically stable with various agents. Using these protoplasts, mushroom breeding is practiced either by transformation or by protoplast fusion. Mushroom breeders of present day are interested in improvement of various parameters of the mushroom strains such as yield, size, colour, flavour, shelf- life, heat and water stress, nutritional parameters, fruiting cycle regulation, sexual compatibility, strain stability and substrate utilization following protoplast technology.
In protoplast based transformation programmes different constructed vectors for high level expression of heterologous genes are used. The resulting constructs are then incorporated into the host system using Agrobacterium mediated transformation or by electroporation. The other method of bringing gene pools of two different mushrooms together is protoplast fusion.
The lack of cell wall allows the plasma membrane of two or more protoplasts to come into intimate contact, something which is not possible under normal circumstances. The sequence of fusion events requires approach, adhesion, lipid-lipid interaction cytoplasmic bridge formation and fusion. Fusion of protoplasts is possible between interspecific, inter generic and even inter-order level members of mushroom but the stable fusants are mostly derived in the fusion of closely related species. The uptake of isolated cell organelles by protoplasts may allow the exploitation of the protoplast fusion technique on a more refined level.
ADVERTISEMENTS:
Ganoderma lucidum (commonly known as Reishi mushroom) is a very important medicinal mushoorm and widely used in China. It has strengthening effects on constitution and health. A vector was constructed for high level expression of heterologous genes in G. lucidum.
The 1.4 kb regulatory region of the glyceraldehyde-3-phosphate dehydrogenase gene (GPD) was isolated from the genomic DNA of Lentinus edodes, and the GPD promoter was fused to the β-glucuronidase (GUS) and bialaphos resistance (bar) genes. Using the resulting construct p301 – bG1, an efficient transformation system based on electroporation was established for G. lucidum. GUS expression was observed among transformants conferring bialaphos resistance.
Different species of Pleurotus are popular edible mushrooms. Somatic hybrids of inter-compatible and inter-incompatible strains were obtained by protoplast fusion. The fusion products between compatible strains P. ostreatus and P. florida formed heterokaryons. Fusants between incompatible strains such as P. cornucopiae + P. florida, P. ostreatus + Ganoderma applanatum formed synkaryons that retained genes from both parents.
The somatic hybrids between compatible strains produced fruiting bodies rapidly and abundantly. Fruiting bodies of the heterokaryans showed intermediate or mixed characters between parents. Fusion products between incompatible strains grew less vigorously than those between compatible strains. Genetic variations within or among inter-compatible and inter-incompatible somatic hybrids of Pleurotus were assessed using RAPD markers.
Single Cell Protein (SCP):
Much of the world population (6-4 billion) is poorly nourished, and famines are frequent. The dietary component that is most usually in short supply is protein. Plants have relatively low protein content and also have very less methionine and tryptophan the essential amino acids. Animals convert plant biomass into high grade proteins but with poor efficiency and the process is time- consuming, so meat is too expensive for much of the world’s population.
Many of the microorganisms are able to convert the cheap sources into nitrogen and ultimately behave as rich sources of proteins. Their rate of growth is also very high and also can be manipulated genetically very easily. These considerations have led to massive industrial development and research aimed at production of microbial biomass for human consumption. Since the main objective is supplying proteins, the biomass is usually termed ‘Single Cell Protein’ or SCP.
In the UK the mitosporic fungus Fusarium graminearum is used for SCP manufacture for human consumption. Use of this fungus is a recent innovation in food technology by the development of Quorn-mycoprotein(R) by a company named Rank Hovis McDougall PLC and ICI (now Zeneca).
The strain was originally isolated from a field in Marlow, Buckinghamshire. The filamentous nature of the biomass is responsible for the meat-like texture and appearance of the final product. It is now sold chiefly as a healthy meat analogue, e.g., sausages and burgers.
Quorn is probably the most thoroughly tested food ever to appear on supermarket shelves, annual sales of which are in excess of 74 million £ in the UK in 1998 with a production of 562 tonnes. SCPs are also produced from the biomass of Candida utilis from a cheap source like molasses, and using hydrocarbon by Saccharomycopsis lipolytica. Introduction of new SCP encountered some problems like safety, acceptability and profitability.
(iii) Food Processing by Fungi:
We have seen that fungal biomass can be used in the form of mushroom or SCP, for food. Fungi also have a crucial role in the processing of many foods, improving the texture, digestability, nutritional value, flavour or appearance of raw materials used.
Some examples are discussed here:
Bread:
The first loaves were probably made about 6,000 years ago, when cereal food were extensively used by humans. The bread making process depends on the conversion of sugar into alcohol and CO2 by yeast. The released CO2 cause the dough to rise giving lightness to the bread whilst the alcohol is driven off during baking. Today about 1.5 million tonnes of baker’s yeasts are exploited annually in the baking industries throughout the world.
Soybean Products:
The soybeans have a high protein content and also yield an oil useful for cooking and many other purposes. Contrary to these the bean also contain a variety of unpleasant compounds, some toxic and some causing excessive gas production in the gut. Cooking destroys some of these factors but a wide range of fermentations developed in China and Japan yield harmless, nutritious and palatable products from soybean.
Soy sauce:
Soy sauce is a condiment extensively used in China, Japan and Indonesia to render a monotonous diet more appetizing. Soya beans are soaked for 16 hours and, after the seed coats have been removed, are cooked. An aerobic, solid substrate fermentation follows in which an appropriate strain of Aspergillus grows on and within the substrate, the mixture of substrate and fungus being known as koji. Inoculation is with spores of either A. oryzae, A. tamari or A. sojae.
The Aspergillus produces a wide range of hydrolytic enzymes, breaking down proteins, polysaccharides and other components of the substrate. This stage continues for 3 days in modern controlled plants. The koji stage is terminated when adequate mycelial growth and enzyme activity has occurred but before sporulation gives mouldy flavours.
The material is then mixed with brine having a salt concentration of about 20%, and the moromi or salt mash stage follows. This is an anaerobic fermentation in deep unstirred vats, the organisms involved being salt-tolerant yeasts and lactic acid bacteria. Pure culture of Zygosaccharomyces rouxii and the lactic acid bacterium Pediococcus can be used to initiate the moromi stage. They also add flavour and aroma in it. The moromi stage lasts at least 3 months and in some processes up to 3 years. At the end of the fermentation the liquid is drained from the remaining solids, clarified, pasteurized and bottled.
Miso:
Miso is a fermented soya bean paste produced mainly in Japan. It has many culinary uses including the preparation of soups and sauces. Usually the main raw materials are soya beans and rice. The production process has much in common with that for soy sauce, with koji and moromi stages. The rice is soaked, steamed and inoculated with Aspergillus oryzae, and a solid substrate fermentation conducted for two days.
It is then mixed with soaked, steamed soya beans, the components of which, like those of the grain, are acted upon by the koji enzymes. Salt is then added and the moromi stage is initiated either with pure cultures of Zygosaccharomyces rouxii or with a starter of fully fermented miso. Liquids are drained from the miso as fermentation proceeds, and these can be used as a sauce.
When a high proportion of rice is used, the moromi stage lasts about 2 weeks, and the final product is light coloured, sweet and not very salty. When a high proportion of soya beans is used, the moromi state lasts longer, and the product is dark, meat-flavoured and salty. Miso is consumed in sufficient amounts to be nutritionally important.
It is a good source of vitamins and proteins, the latter being in a readily assimilated form and high in some amino acids, such as lysine, which are often deficient in predominantly vegetarian diets.
Other important soybean fermented products are Tempe (or tempeh)—an Indonesian fermented food fermented by pure cultures of Rhizopus oligosporus and Sufu fermented by Mucor racemosus, Rhizopus chinensis and Actinomucor elegans.
(iv) Cheese and Fermented Milk:
The preparation of cheese and fermented milks such as yoghurt is a fermentation industry second only to brewing in scale and in the value of its products. The main microorganisms involved are lactic acid bacteria. However, fungi have an important but subsidiary role in the production of some cheeses and fermented milks, particularly during the process of ripening.
In cheese production, milk is inoculated with starter cultures of lactic acid bacteria. The enzyme rennin is then added to coagulate milk proteins and form a curd. Here there can be indirect fungal involvement, as there is an increasing use of rennin from fungi instead of calf rennet.
The way the curd is then handled depends on the type of cheese being made, but with all cheeses whey has to be drained from the curd. Various microorganisms in addition to lactic acid bacteria have a role in subsequent cheese ripening, and instances where fungi make a major contribution will be considered below.
Surface-Ripened Cheese:
The best-known surface-ripened cheeses are Brie and Camembert. The curd is drained and shaped to give the final disc or segmented disc form. The surface is then dry-salted and sprayed with spores of the white Penicillium species P. camemberti. Salt and acid-tolerant yeasts and the mitosporic fungus Geotrichum candidum develop at the surface although deliberate inoculation is not practiced.
It is thought that utilization of lactic acid by the yeasts may raise the surface pH and encourage the germination of Penicillium spores and subsequent mycelial growth. The white mycelium gives the surface of Camembert and Brie its characteristic appearance but does not penetrate the interior. Deamination of amino acids by Geotrichum produces traces of ammonia and contributes to flavour. The Penicillium makes lipases which release fatty acids, which can be converted into methyl ketones, both of which contribute to flavour, as do a range of other fungal products.
Blue-vein cheese:
The best-known blue-vein cheeses are Roquefort, Gorgonzola, Stilton and Danish Blue. The starter bacteria include a heterolactic fermenter which produces carbon dioxide as well as lactic acid. The gas production results in irregular cavities in the cheese. An inoculum of Penicillium roqueforti spores is either included in the starter or added to the fresh curd.
After the curd has been compressed into a cheese, salt is dusted on to the surface and diffuses into the interior, establishing a concentration gradient. The cheese is then spiked, and the air that enters allows the germination of Penicillium spores, and mycelial growth along the perforations and through the cavities in the cheese. Growth is greatest at the intermediate salt concentrations at moderate depth in the cheese, and it is there that the blue coloration resulting from the production of P. roquefortii spores is most marked. The fungus produces both proteolytic and lipolytic enzymes, resulting in the release of amino acids and peptides from protein and fatty acids from lipid.
The fatty acids to some extent undergo β-oxidation which is a normal part of fatty acid catabolism. This, by removing acetyl units, results in fatty acids of shorter chain length. These fatty acids can be oxidized to the corresponding β-keto acid and decarboxylated to a methyl ketone. Heptan-2-one is the major methyl ketone in blue- vein cheese, in contrast to the C9 and C11 methyl ketones in the surface ripened cheeses such as Camembert. Fatty acids, methyl ketones and lactones are all-important in determining the flavour of blue-vein cheese.
B. Fungal Secondary Metabolites:
(i) Antibiotics:
Of the several thousand antibiotics discovered till date, about 22% could be produced by filamentous fungi such as Penicillium chrysogenum, Cephalosporium sp. etc. The outstanding performance of the present days P. chrysogenum was achieved after a series of mutation from its original isolate NRRL1951.
Today most penicillin is produced with this spicies, grown in aerobic stirred fermenters, which gives 55-fold higher penicillin yields than the original static culture of P. notatum. At present, natural penicillin G and the biosynthetic penicillin V, have a combined market of $ 4.4 billion, and many semisynthetic penicillin’s and cephalosporin’s, which have a market of $ 11 billion.
(ii) Immunosuppressive Agents:
Cyclosporin A was originally discovered as a narrow spectrum antifungal peptide produced by the mold Tolypocladium nivenum. Discovery of the drugs immunosuppressive activity led to its use in heart, liver and kidney transplants and thus to the overwhelming success of the organ transplant field.
A very old broad spectrum fungal antibiotic, mycophenolic acid, produced by some species of Penicillium, was never commercialized as an antibiotic, but its 2-morpholinoethylester was approved as a new immunosuppressant for kidney transplantation in 1995 and for heart transplants in 1998.
(iii) Cholesterol Lowering Drugs:
Commercially, the most important group of these compounds are the mevinic acids, which are potent cholesterol-lowering agents. The fungal statins like lovastatin and compactin are important to be mentioned. These are produced by Penicillium citrinum and Monascus ruber.
These components inhibit 3-hydroxy- 3-methylglutaryl-CoA reductase an enzyme responsible for regulatory and rate limiting function for cholesterol biosynthesis in liver. Lovastatin was approved by FDA (Federal Drug Agency) in 1987. This component along with other such agents have a market of $ 15 billion.
(iv) Antitumor Agents:
Taxol, a natural anticancer agent, was originally discovered in plants but can also be produced by the fungus Taxomyces andreanae. It is approved for the treatment of breast and ovarian cancer and is the only commercial antitumor drug known to act by blocking depolymerization of microtubules. Coriolus versicolor, a basidiomycetous fungus also produces anticancerous principles which kill neoplastic cells by activating NK-cells.
(v) Mycotoxins as Medicine:
At least a dozen of ergot alkaloids produced from an ascomycetous fungus Claviceps purpurea are used in medicine. Ergometrine and its derivatives act upon the uterine smooth muscles, contract them and facilitate delivery. They are also given after childbirth to prevent excessive bleeding. They control this by narrowing blood vessel diameter. Ergotamine and its derivatives are used to treat migraine.
Here again the alkaloids acts through vasoconstriction, counteracting the dilation of veins in the brain that occur in migraine. Bromocryptine, a derivative of a naturally occurring ergocryptine, is used to stop excessive or untimely milk production.
It does this by suppressing the prolactin formation from pituitary gland, the hormone that stimulates lactation. Zearelanone, produced by Gibberella zeae is an estrogen and its reduced derivative zeranol is used as an anabolic agent in cattle and sheep, increasing both growth and feed efficiency.
(vi) Pigments:
Food grade dyes with medicinal values are obtained from Monascus purpureus. Koji, also called red rice, is a Chinese food is produced from fermentation of M. purpureus. The water-soluble red pigments monascorubramine and rubropunctamine are used for preparing wine, soybean cheese, meat etc. along with red rice and is authorized for food used in China and Japan. Another orange coloured carotenoid astaxanthin from yeast Phaffia rhodozyma is used for coloring salmonid flesh and crustaceous shells.
(vii) Polyunsaturated Fatty Acids:
Morteriella isabellina and Mucor circinelloides can accumulate up to 5 g/l linoleic acid in a medium based on molasses or glucose. Morteriella alpine is the best choice for the production of arachidonic acid. Synthesis of this polyunsaturated fatty acid is required for the cloning of all desaturases.
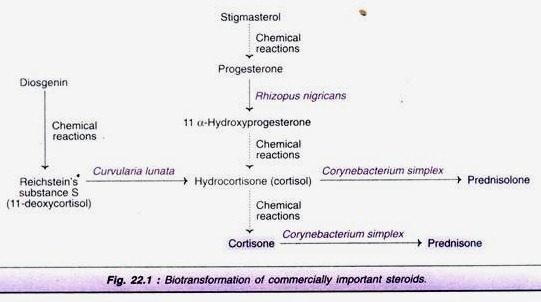
(viii) Steroid Transformation:
The Zygomycete Rhizopus nigricans carries out an 11 α-hydroxylation and the mitosporic fungus Curvularia lunta an 11 β-hydroxylation on the steroid molecule to produce 11 α-hydroxyprogesterone and hydrocortisone, respectively. These components are therapeutically very useful.
C. Fungal Enzymes:
The mediation of chemical reactions by catalytic proteins (enzymes) is a central feature of living systems. Living cells make enzymes although the enzymes themselves are not alive. We can encourage living cells to make more enzymes than they would normally make or to make a slightly different type of enzyme (protein engineering) with improved characteristics of specificity, stability and performance in industrial processes. These enzymes usually operate under mild conditions of pH and temperature.
A wide range of enzymes are excreted by fungi and play an important role in the breakdown of organic materials ; many of these enzymes are now produced commercially.
D. Bio Control Agents and Other Uses:
Fungi possess a number of characteristics that make them potentially ideal bio control agents. Firstly, many saprophytic species antagonize, representatives of all the pest organisms, including plant pathogenic fungi, weeds and insects. Secondly, fungi can be readily grown in culture so that large quantities can be economically produced for release, mainly as spores or mycelial fragments, into the environment.
These inoculants then germinate or grow to produce active mycelium which can parasitize or otherwise inhibit the pest without damaging the non-target organisms. Fungi also survive for relatively long periods as resting bodies, and can then germinate to grow and control the target population thereby making continual re-inoculation with the bio-control agent unnecessary.
Example of Bio Control Agents Used Commercially:
1. Trichoderma harzianum—White rot onion
2. Phlebia gigantean—Heterbasidion root rot pine
3. Agrobacterium radiobactor var. radiobactor—Crown gall rose
4. Sporidesmium sclerotivorum—Lettuce drop lettuce
5. Talaromyces flavus—Damping off sugarbeet.
Use of Fungi to Control Insect Pest:
Over 400 species of fungi attack insects and mites, so there is great potential for the use of these organisms as biological insecticides. As insect bio control agents, fungi are markedly superior to other microorganisms because they are generally non-specific in their action and are useful against a wide range of insect pests.
Most of the so-called entomopathogenic fungi are phycomycetes and Deuteromycetes. Spores of these fungi attack either the external or gut cuticle of their insect hosts. They then germinate and hyphae penetrate the haemocoel. Death may result from the production of a toxin by the fungus.
Use of Fungi to Control Nematodes:
Fungi that parasitise nematodes (nematophagus fungi) can be divided into nematode- trapping fungi, endoparasitic species and fungi that parasitise nematode eggs. Nematode trapping fungi capture nematodes with specialized structures such as constrictive and non-constrictive rings, adhesive knobs or, lastly, by producing an adhesive material along the entire mycelial surface. Endoparasitic nematophagous fungi live in soils where they produce adhesive spores.
These become attached to body of the nematode, on germination, a germ tube enters the body where it grows and consumes the host. Egg parasites, as their name suggests, are nematophagous fungi that parasitise the eggs of nematodes.
The following approaches have been used in an Attempt to use Fungi to Control Nematodes:
(1) Exploitation of naturally suppressive soils.
(2) Soil amendments to encourage indigenous bio-control agents.
(3) Inoculation of soils with selected strains of bacterial & fungi.
(4) The use of microbial toxins & enzymes.
E. Application of Molecular Biology in Fungal Biotechnology:
Apart from selection of potential fungal strain by screening or improvement of strains by conventional mutagenesis like UV, or chemical mutagenesis, molecular techniques have opened new avenues to use yeasts and molds as cell factories for the production of our desired proteins or compound.
Some Important Products Obtained by Recombinant DNA Technology are:
(i) Pharmaceutical Proteins:
Since it is a food organism, Saccharomyces cerevisiae is considered to be a safe host for the production of pharmaceutical proteins. This yeast can be grown rapidly and to high cell density, can secrete heterologous proteins into the extracellular broth, and knowledge of its genetics is more advanced than that of any other eukaryote.
Its complete genome has been sequenced in 1996. Mammalian genes like human epidermal growth factor, human interferon and human hemoglobin genes have been cloned and expressed in S. cerevisiae. The most commercially important yeast recombinant process has been the production of genes encoding surface antigens of the Hepatitis B virus, resulting in the first safe hepatitis B vaccine ‘engerix’ and ‘recombivax’.
Although S. cerevisiae possesses many such successful biotechnological applications, sometimes it is also regarded as a less effective host for large scale production of mammalian proteins because of certain drawbacks viz., hyperglycosylation and the absence of strong and tightly regulated promoters. To overcome these drawbacks scientists worked a lot. It has been found that Pichia pastoris — a methylotrophic yeast—has a very powerful and extensively used expression systems.
The Avantages of this Oganism are:
(i) An efficient and tightly regulated methanol promoter (AOX1) which yields alcohol oxidase at 30% of soluble protein,
(ii) Low rate of glycosylation, due to shorter chain lengths of N-linked high mannose oligosaccharides, usually up to 20 residues lacking the terminal α-1,3 mannose linkages,
(iii) Intergration of multiple copies of foreign DNA into chromosomal DNA yielding stable transformants,
(iv) Secretion of high level of foreign proteins,
(v) High density growth and straight-forward scale up.
A good number of recombinant proteins of both intracellular and extracellular nature have been produced from P. pastoris. In spite of its very excellent expression system, the only drawback of this beautiful organism is its non-GRAS status, although some products made by this yeast are being evaluated in phase-III clinical trials. For example, production of recombinant hirudin — which is a thrombin inhibitor from the medicinal leech Hirudo medicinalis, results in yields of 1.5 g/l secreted product.
Hansenula polymorpha, another member of methylotrophic yeast, also possesses a strong expression system like P. pastoris. The promoter of the methanol oxidase gene is used to express foreign genes. Like AOX1 of P. pastoris, MOX in H. polymorpha is also highly expressed and tightly regulated, giving enzyme levels up to 37% of total cell protein.
The only difference of this system is that its expression is significantly derepressed during glucose limitation and, therefore, tight regulation of the MOX promoter is lost under the high glucose conditions usually used for high biomass fermentations.
The development of molecular techniques in filamentous fungi for the production of recombinant heterologous proteins is laborious and has contrasted markedly with the success achieved in yeasts. Agrobacterium tumefaciens Ti-plasmid mediated transformation and restriction enzyme mediated integration are certain advances in this regard. Production of heterologous proteins are lower than the levels of fungal proteins.
This is due to the factors that influence production i.e., transcription, translation, secretion, and extracellular degradation. Many strategies have been developed to overcome these problems, including the construction of protease deficient mutants, the introduction of a large copy of genes, the use of strong fungal promoters, efficient secretion signals, and fusion with a gene that encodes part of or an entire well secreted protein. Gene fusion is the first choice in attempting to produce heterologous proteins in fungal hosts. Fusion has resulted in secretion of 5 mg/l human interleukin-6, 2 mg/l human lysozyme and 250 mg/l human lactoferrin.
Recent studies have shown that the fungal secretory pathway is the most important limiting factor for heterologous protein production. Screening of mutants with altered secretion properties using green fluorescence protein as reporter, elucidation of the role of secretion related chaperones, kinetic studies on protein secretion, and the effect of hyphal branch frequency are examples of the work being carried out to understand this complex process.
N-glycosylation is important for many pharmaceutically important proteins for stabilization and proper folding. Although O-linked glycosylation in yeast is quite different from that in higher eukaryotes, N-linked glycosylation is more conserved. Glycosylation of a protein can be different depending on factors such as the medium in which they are grown.
(ii) Commercial Recombinant Enzymes:
Recombinant fungi are one of the main sources of enzymes for industrial applications. The industrial enzyme market reached $ 2.0 billion in 2000 for the following application areas: food 45%, detergents 34%, textiles 11%, leather 3%, pulp and paper 1.2%. This does not include therapeutic and diagnostic enzymes.
Most of these enzymes used in detergent, food and starch processing industries are recombinant products. Although the number of heterologous fungal enzymes approved for food applications are not very large, the list is continuously increasing.
One of the heterologous protein, calf reinin (chymosin), is expressed in Aspergillus niger var. awamori and is used for cheese making. Microbial lipases have a huge potential in areas such as food technology, biomedical sciences and chemical industries since they are:
(i) Stable in organic solvents,
(ii) Possesses broad substrate specificity,
(iii) Do not require cofactors, and
(iv) Exhibit high enantioselectivity.
In the food industry, lipases are commonly used in the production of fruit juices, baked foods, desirable flavours in cheeses, and interesterification of fats and oils to produce modified acylglycerols. Three fungal recombinant lipases are used currently in the food industry—Rhizomucor miehi, Thermomyces lanuginosus and Fusarium oxysporum, all of which are produced in A. oryzae.
Lipases are extremely important in the detergent industry. They are extensively used in household detergents, industrial cleaners and leather processing where they can be combined with proteases, oxidases and peroxidases.
To be suitable, lipases should be alkalophilic, able to work above 45°C and at pH values of 10, and capable of functioning in the presence of various oxidants and surfactants. Novo Nordisk introduced the first commercial recombinant lipase for use in a detergent, by cloning the Humicola lanuginose lipase gene into the A. oryzae genome.
(iii) Strain Improvements:
Apart from classical mutagenesis by radiations or chemical mutagens of the industrially important fungal strains, application of recombinant DNA- technologies is of immense importance. Continued progress in the area of metabolic engineering has led to overproduction of limiting enzymes of important biosynthetic pathways, thus increasing production of final products. Brewing yeasts have been engineered by cloning endoglucanase from Trichoderma reesei to it. This enzyme hydrolyzes barley glucans and makes the beer clean.
Similarly, starch utilizing S. cerevisae strains produce lower acidity and enhanced flavour. Brewing yeasts also have been engineered to produce acetolactate decarboxylase from Enterobacter aerogenes and Acetobacter aceti. This enzyme eliminates diacetyl and the requirement of 4-week flavour maturation is reduced to 1-week fermentation stage. The resulting beer suffers no loss of quality and flavour.
Replacement of the native promoter of the ACVS-encoding gene in A. nidulans increased penicillin production 30-times. Expression of Cef E from Streptomyces clavuligerus in P. chrysogenum led to recombinant strains able to produce cephalosporin intermediates.
Thaumatin, a protein from the bacteria Thaumatococcus danielli with an intense sweetness (about 3,000 times more than sucrose), has been recently approved as a food grade ingredient. Successful expression of thaumatin was achieved in Penicillium roquefortii and A. niger var awamori at titers of 2-7 mg/lit. Recently this titer has been increased to 14 mg/lit by using stronger promoters and gene doses in A. niger var awamori.
Production of β-carotene in the food grade yeast Candida utilis—containing the biosynthetic genes from bacteria Erwinia uredovora—is in progress. Using similar cloning strategy, recombinant yeast strains are developed to produce α-linoleic acid from two desaturases of Morteriella alpina.
(iv) Entry of DNA into Fungi:
DNA can be introduced into fungi in a variety of ways. Fungi possess tough cell walls which block direct uptake of DNA, necessitating either removal of the cell wall (protoplast formation) or forcible injection of DNA into the cell by particle bombardment electroporation or Agrobacterium-mediated transformation.
Traditional Methods have Relied on PEG-Stimulated Uptake of DNA by Protoplasts but More Recent Developments are:
Protoplast Formation:
To Isolate Protoplast from Fungal Cells the following Steps are to be followed:
(i) Enzymatic Digestion of Fungal Cell Walls:
Enzymatic digestion of the complex polysaccharide, chitin cell wall of fungi results in blabbing of cytoplasm and release of spherical protoplasts. Protoplasts contain fungal cytoplasm and are heterogeneous. With some containing single nuclei, others multiple nuclei, and some anucleate.
The commercially available enzyme mixtures which are most suitable for fungal protoplast formation are Novozyme 234 and Glucanex, which are both derived from mycolytic Trichoderma species. Where the action of these enzymatic preparations mixtures is not sufficient to produce sufficient numbers of protoplasts, small amounts of helicase, driselase, or other lytic enzymes may need to be added. In general, 5-10 mg/ml of Novozyme or Glucanex is sufficient to produce usable numbers of protoplasts.
(ii) Osmotic Stabilization of Protoplasts:
Protoplasts will immediately lyse unless they are released into osmotically-stabilized medium. Typical media for osmotic stabilization consist of roughly 1 M salt or sugar solutions. Common osmotic stabilizers include 0.6-0.8 M KCl, 1-1.2 M MgSO4, 0.8-1.2 M NaCl, and 1 M sucrose. These are added to conventional media for fungal propagation.
(iii) Starting Material:
Not all ages or types of mycelium will yield protoplasts with equal efficiency. A rule-of-thumb is to use hyphae that are as young as possible — ideally very young hyphae that have just germinated from spores should be prepared. An overnight culture, or two-day-old culture of most ascomycetes, or badsiomycete fungi is sufficient for effective protoplast preparation.
Using the Protoplasts the Incorporation of DNA can be done by the following methods:
(a) REMI Transformation:
Restriction enzyme-mediated integration (REMI) has been used as a means of introducing DNA into fungi for mutagenesis. Addition of restriction enzymes has also been used to facilitate integration of transforming DNA. Restriction enzymes are included with transforming DNA and are thought to facilitate integration by nicking chromosomal DNA at their cognate recognition site.
This technique has been widely used for production of insertional mutants as the transforming DNA integrates in the genome in a predictable manner and can be easily rescued from mutants of interest. Many enzymes have been found that increase transformation from several- fold to a hundred-fold.
These include four base pair recognizing enzymes and six base pair recognizing enzymes. The increase (or decrease) in transformation frequency is dependent on the amount of enzyme included with the transforming DNA which must be determined empirically.
(b) Agrobacterium-Mediated Transformation of Fungi:
Agrobacterium tumefaciens is a soil bacterium, which has the ability to transfer sections of plasmid borne DNA to plants. This ability has long been exploited for DNA-mediated transformation of higher plants. A. tumefaciens has recently been shown to transfer DNA to fungi in a similar manner.
This method of transformation is potentially simpler and more reliable than standard methods, although it has not been proven in many species to date. DNA for transformation, including a selectable marker and any genes of interest, must first be cloned between left and right T-DNA borders in a binary transformation vector such as pBIN19.
This vector must then be transferred into A. tumefaciens. Bacteria containing the DNA of interest are then incubated with fungus to allow DNA transfer, before being killed by antibiotic treatment.
(c) Particle Bombardment:
Microprojectile bombardment involves projecting DNA coated gold or tungsten particles into intact cells. Over the last few years, micro projectile bombardment has become a powerful tool for transformation of intractable plant species, such as the commercial cereals. Several filamentous fungi have been transformed by means of microprojectile bombardment. This technique is particularly useful for transformation of fungi which cannot be cultured outside their host, such as obligate biotrophs.
F. Future Prospects:
The last few years have been a period of great progress using fungi as cell factories. There are four major fronts in which work is currently underway. The first is the development of alternative hosts, especially those that have already been given GRAS status by the FDA and can be used in the food industry. Research is being focused on species such as Aspergillus sojae, Aspergillus japonicus, Mortierella alpina and Fusarium veneratum, among others.
The second front is the development of better molecular techniques to improve expression and secretion of non-fungal proteins in filamentous fungi. The third major front involves the use of these molecular techniques to carry out metabolic engineering in order to modify and improve particular biosynthetic pathways.
The final front will utilize the techniques dealing with the overall analysis of gene expression, i.e., genomics, proteomics and metabolomics. Four fungal genomes have already been sequenced—S. cerevisiae, Schizosaccharomyces pombe, A. niger , Neurospora crassa and sequencing of four others are in progress. Initial steps on filamentous fungal genomics and proteomics have recently been published and undoubtedly much more will become available in the years ahead.
The future of fungal biotechnology is encouraging when one considers that all the contributions that have been made already by fungi have been done with less than 5% of the fungal species present in nature. Soils and marine environments contain thousands of unknown microbial species, many of them fungi.
New methods are being used to harness “environmental DNA” and to bring about the cultivation of so-called ‘unculturable’ microorganisms. About 30-50% of known proteins have no known function. As more functions are revealed by functional genomics and bioinformatics, new targets will become available for screening fungal products. Fungal enzymes will be improved in activity, specificity, and stability by directed evolution.
Secondary metabolite pathways of fungi will be enhanced by directed evolution of whole cells (“whole genome shuffling”) in concert with metabolic engineering. New secondary metabolites will be created by combinational biosynthesis in fungi.