ADVERTISEMENTS:
Read this article to learn about the scale-up process in the animal cell culture system.
The scale-up process is divided into two categories. The two categories are: (1) Scale-up in Suspension and (2) Scale-up in Monolayer and also about the monitoring of cell growth in the scale-up process.
Scale-Up Process:
Scale-up involves the development of culture systems in stages from (small scale) laboratory to (large scale) industry. The methodology adopted to increase the scale of a culture depends on the proliferation of cells and is broadly divided into two categories.
ADVERTISEMENTS:
1. Scale-up in suspension.
2. Scale-up in monolayer.
Scale-Up in Suspension:
Scale-up in suspension is the preferred method as it is simpler. Scale-up of suspension culture primarily involves an increase in the volume of the culture. Small scale generally means the culture capacity less than 2 litres volume (or sometimes 5 litres).
Stirred suspension cultures:
ADVERTISEMENTS:
It is usually necessary to maintain cell strains in stirred suspension cultures, by agitation (or stirring) of the medium. The stirring of the culture medium is achieved by a magnet encased in a glass pendulum or by a large surface area paddle. The stirring is usually done at a speed of 30-100 rpm. This is sufficient to prevent sedimentation of cells without creating shear forces that would damage cells.
Static suspension cultures:
Some cells can grow in suspension cultures, without stirring or agitation of the medium, and form monolayer cells. However, static suspension cultures are unsuitable for scale-up.
Factors in Scaling-Up:
For appropriate scale-up, the physical and chemical requirements of cells have to be satisfied.
Physical parameters:
i. Configuration of the bioreactor.
ii. Supply of power.
iii. Stirring of the medium.
ADVERTISEMENTS:
Chemical parameters:
i. Medium and nutrients.
ii. Oxygen.
iii. pH and buffer systems.
ADVERTISEMENTS:
iv. Removal of waste products.
Some of the relevant aspects of the factors in scale-up have already been described under cell culture-general considerations The most commonly used techniques for stirred suspension cultures are briefly described hereunder.
Stirrer Culture:
A diagrammatic representation of a stirrer flask is shown in Fig 37.2. The size of the stirrer flask is in the range of 2-10 litres. It is fitted with a magnetized rotating pendulum, and two side arms — one for the addition of cells and medium, and the other for the supply of CO2.
The stirrer culture vessel is autoclaved (at 15 lb/ in2 for 15 minutes), and is then set up as in Fig. 37.2. The flask is seeded with the culture. Then medium along with an antifoam agent is added. The flask is connected to CO2 and stirred at a speed of 60 rpm. The flask is incubated for about 2 hours.
The contents of the small stirrer flask are transferred to a large flask and the entire set up is restarted. Incubation at 37°C is carried out for 4-7 days. The growth of the cells is monitored daily, and the cells are counted. There is a tendency of the cells to enter apoptosis, if the concentration exceeds 1 × 106 cells/ml.
Continuous Flow Culture:
In a continuous flow culture, it is possible to keep the cells at a desired and set concentration, and maintain. This is carried out by a bio-stat or chemo-stat (Fig 37.3).
Continuous flow culture consists of growing the cells at the mid-log phase, removal of a measured volume of cells, and replacement by an equal volume of medium. The equipment, specially designed for this purpose has the facility for removal of the cells and addition of medium.
The flow rate of the medium addition can be determined from the growth rate of the culture. The medium flow can be regulated by a peristaltic pump. By this technique, it is possible to keep the culture conditions constant rather than to produce large number of cells. The continuous flow cultures are useful for monitoring metabolic changes in relation to cell density. However, these cultures are more susceptible to contamination.
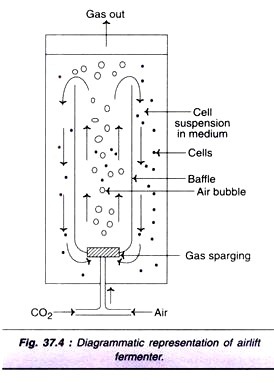
Air-Lift Fermenter Culture:
The major limitation of scale-up in suspension culture is inadequate mixing and gas exchange. For small cultures, stirring of the medium is easy, but the problem is with large cultures. The design of fermenter should be such that maximum movement of liquid is achieved with minimum shear to damage the cells.
A diagrammatic representation of an air-lift fermenter is depicted in Fig. 37.4. A 5% CO2 in air is pumped through the bottom of the fermenter. The bubbles formed move up to agitate and aerate the culture. These bubbles carry a flow of liquid along with them and release at the top which goes to the bottom for recycling.
It is possible to continuously supply O2 to the culture in this technique. Air-lift fermenter culture technique is suitable for fragile animal as well as plant cells. This fermenter is extensively used in the biotechnology industry for culture capacities up to 20,000 litres.
ADVERTISEMENTS:
NASA bioreactor:
NASA (National Aeronautics Space Administration, USA) constructed a bioreactor to grow the cells at zero gravity by slowly rotating the chamber (Fig. 37.5). The cells remain stationary and form three-dimensional aggregates and this enhances the product formation. In the NASA bioreactor, there is almost no shear force; hence the cells are not damaged.
As the culture chamber stops its rotations, the cell aggregates sediment and the medium can be replaced.
Other Systems for Suspension Culture:
ADVERTISEMENTS:
Rotating chambers:
The mixing and aeration of the culture medium can be achieved by 2 or 3 rotating chambers. The chambers are so designed that the cell suspension and mixing are high in one chamber while the product and spent medium remain in the other chamber. These chambers are separated by semipermeable membrane.
Perfused suspension culture:
This also has two compartments. The cells are kept in a low-volume compartment at high concentration, while the medium is perfused in adjacent compartment. The product can be collected in a third compartment.
Scale-Up in Monolayer:
The monolayer culture are anchorage- dependent. Therefore, for the scale-up of monolayer cultures, it is necessary to increase the surface area of the substrate in proportion to the number of cells and volume of the medium. Suspension cultures are preferred as they are simple. The advantages and disadvantages of monolayer cultures are listed.
Advantages:
i. Change of medium and washing of cells easy.
ii. It is easy to perfuse immobilized monolayer cells.
iii. The cell product formation (pharmaceutically important compounds e.g. interferon, antibodies) is much higher.
iv. The same set up and apparatus can be repeatedly used with different media and cells.
Disadvantages:
i. Tedious and costly.
ii. Require more space.
iii. Growth of cells cannot be monitored effectively.
iv. Difficult to measure control parameters (O2 pH, CO2 etc.)
For scale-up of monolayer cultures, a wide range of tissue cultures and system have been developed. A selected few of them are briefly described.
Roller Bottle Culture:
A round bottle or tube is rolled around its axis (by rollers) as the medium along with the cells runs around inside of the bottle (Fig. 37.6). As the cells are adhesive, they attach to inner surface of the bottle and grow forming a monolayer.
Roller bottle culture has certain advantages.
i. The medium is gently and constantly agitated.
ii. The surface area is high for cell growth.
iii. Collection of the supernatant medium is easy.
There are limitations in roller culture.
i. Monitoring of cells is very difficult.
ii. Investment is rather high.
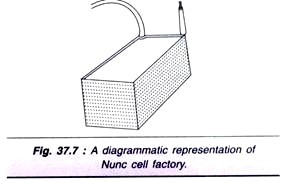
Multi-surface Culture:
The most commonly used multi-surface propagator of monolayer is Slunclon cell factory (in short Nunc cell factory). It is composed of rectangular petri dish-like units with huge surface area (1,000-25,000 cm2). The units are interconnected at two adjacent corners by vertical tubes (Fig. 37.7). The medium can flow between the compartments from one end.
The cell factory is almost like a conventional petridish or a flask with multiplayer units. The main limitation of cell factory is that it is very difficult to monitor the growth of cells. The major advantage however, is its simple operation to produce large number of cells.
Multi-array Disks, Spirals and Tubes:
The surface area for growth of monolayer cultures can be increased by using disks, spirals or tubes. They are however, not in common use as their commercial importance is limited.
Micro-carrier Culture:
Monolayers can be grown on small spherical carriers or micro-beads (80-300 pm diameter) referred to as micro-carriers. The micro-carriers are made up of any one the following materials (trade names given in brackets).
i. Plastic (acrobeads, bioplas).
ii. Glass (bioglass, ventreglas).
iii. Gelatin (ventregel, cytodex-3).
iv. Collagen (biospex, biospheres)
v. Cellulose (DE-52/53).
vi. DEAE Dextran (cytodex I, dormacell).
The micro-beads provide maximum surface area for monolayer cultures. This actually depends on the size and density of the beads. The cells can grow well on the smooth surface at the solid-liquid interface. However, micro-carriers need efficient stirring without grinding the beads. The main advantage with micro-carrier culture is that it can be treated as a suspension culture for all practical purposes.
Micro-carriers can be cultured in stirrer flask (See Fig. 37.2) or in continuous suspension (See Fig. 37.3). In fact, the suppliers of micro-carriers provide the technical literature and other relevant information for setting up a micro-carrier culture.
Factors affecting micro-carrier culture:
i. Composition and coating of beads (gelatin and collagen beads are preferred as they can be solubilized by proteases).
ii. Higher stirring speed is usually required.
iii. Glass beads are used when the micro-carriers need to be recycled.
Analysis of micro-carrier culture:
The cell counting techniques are difficult to be used for micro-carrier cultures. The growth rate can be detected by analyzing DNA or protein.
Perfused Monolayer Culture:
The growth surface areas of the monolayer cultures can be perfused to facilitate medium replacement and improved product formation and recovery. The perfusion can be carried out with pumps, oxygenator and other controllers. Perfusion of fixed and fluid-bed reactor is briefly described.
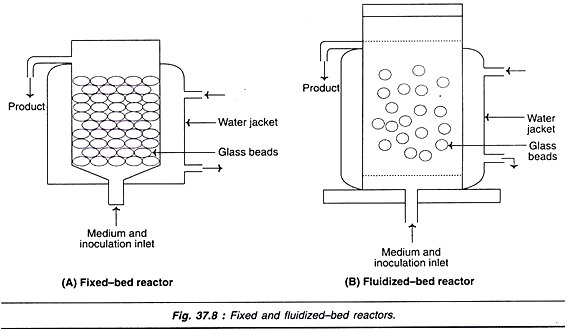
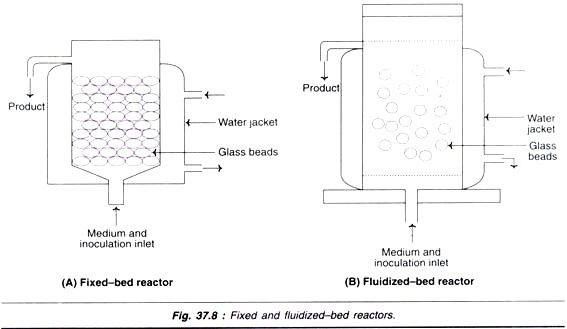
Fixed-bed reactors:
The fixed-bed reactor has a bed of glass beads (Fig. 37.8A). The medium is perfused upwards through the bed. The cells are grown on the surfaces of the beads. The products can be collected from the top along with the spent medium.
Instead of glass beads, porous ceramic matrix with micro-channels can also be used in fixed-bed reactors.
Fluidized-bed reactors:
In a fluidized-bed reactor, the beads are suspended in a stream of medium (Fig. 37.8B). These beads are porous in nature, and are made up of ceramics or a mixture of ceramics mixed with natural products such as collagen. They are of low density and float in the medium. The flow rate of the perfused medium is equal to the sedimentation rate of the beads. The cells can grow as monolayers on the outer surfaces and inside of the porous beads.
Other perfused monolayer cultures:
Membrane perfusion, hollow-fiber perfusion, matrix perfusion and microencapsulation are among the other techniques for perfusion of monolayer cultures.
Monitoring of Cell Growth in Scale-Up:
Monitoring of the progress of cell growth and the culture systems are very important in scale-up.
Monitoring of Suspension Cultures:
The progress of suspension cultures can be monitored in situ by measuring glucose, O2, CO2, pH or metabolites produced (lactate, ammonia) or specialized products formed (e.g. immunoglobulin’s by hybridoma cells).
The cell proliferation and rate of biomass formation can also be determined by estimating DNA, protein and ATP. The different parameters for monitoring of a bioreactor for suspension culture are depicted in Fig 37.9.
Monitoring of Monolayer Cultures:
It is rather difficult to monitor monolayer cell cultures for scale-up. This is due to the fact that in most of the techniques employed for monolayer cultures, the cells cannot be observed directly to monitor the progress of the culture.
In recent years, nuclear magnetic resonance (NMR) technique is used to assay the contents of culture. The characteristic NMR spectra generated by specific metabolites enables the identification and quantitation of metabolites, besides detecting the progress of cell growth.