ADVERTISEMENTS:
In this article we will discuss about Paramecium Caudatum:- 1. Habit, Habitat and Culture of Paramecium Caudatum 2. Structure of Paramecium Caudatum 3. Locomotion 4. Nutrition 5. Respiration and Excretion 6. Osmoregulation 7. Behaviour 8. Reproduction 9. Aberrant Behaviour in Reproduction 10. Some Cytoplasmic Particles.
Contents:
- Habit, Habitat and Culture of Paramecium Caudatum
- Structure of Paramecium Caudatum
- Locomotion of Paramecium Caudatum
- Nutrition in Paramecium Caudatum
- Respiration and Excretion in Paramecium Caudatum
- Osmoregulation of Paramecium Caudatum
- Behaviour of Paramecium Caudatum
- Reproduction in Paramecium Caudatum
- Aberrant Behaviour in Reproduction in Paramecium Caudatum
- Some Cytoplasmic Particles Reported in Paramecium Caudatum
1. Habit, Habitat and Culture of Paramecium Caudatum:
Paramecium caudatum (Gr., paramekes = oblong; L., caudata = tail) is commonly found in freshwater ponds, pools, ditches, streams, lakes, reservoirs and rivers. It is specially found in abundance in stagnant ponds rich in decaying matter, in organic infusions, and in the sewage water. Paramecium caudatum is a free-living organism and this species is worldwide in distribution.
Culture of Paramecium:
ADVERTISEMENTS:
Take submerged weeds from a pond and place in a jar of distilled water, cover the jar and leave it to rot; swarms of Paramecia will appear in a few days. Now boil hay in water, decant the infusion and add a few grains of wheat, and let it stand till turbid with bacteria.
Transfer Paramecia from the first jar into this liquid where they will multiply rapidly. Hay infusions alone will produce Paramecia showing presence of cysts, and cyst resembling sand grains have been reported, but there is no proof of Paramecia forming cysts, since they have never been confirmed.
2. Structure of Paramecium Caudatum:
(i) Size and Shape of Paramecium Caudatum:
Paramecium caudatum (Fig. 20.1) is a microscopic organism and visible to the naked eyes as a minute elongated body.
ADVERTISEMENTS:
It appears light gray or white measuring commonly between 170 to 290 microns in length and may attain a length up to 300-350 microns. P. caudatum looks like the sole of a slipper or shoe, hence, the animal is commonly known as slipper animalcule. It is four times as long as broad and somewhat cylindrical with distinctly different ends.
The forward moving anterior part is slender with a blunt or rounded end, while the posterior end is somewhat pointed or cone-shaped. The widest part of the organism is just below the middle. The body of the animal is asymmetrical in form showing a well defined oral or ventral surface and an aboral or dorsal one.
(ii) Pellicle:
The body is covered by a thin, double layered, elastic and firm pellicle made of gelatin. The pellicle holds the shape of the animal but is elastic enough to permit contractions. The pellicle has double membrane, the outer membrane is continuous with the cilia and the inner membrane with the ectoplasm. Under the higher magnification of microscope, pellicle shows rectangular or hexagonal depressions on its surface.
This arrangement is preserved on the dorsal surface of Paramecium but on the ventral surface the ridges converge in front and behind towards a preoral and postoral aperture. Each hexagonal depression is perforated by a central aperture through which a single cilium emerges out. The anterior and posterior margins of hexagonal depressions bear the openings of trichocysts.
The electron microscopic study of pellicle (Fig. 20.2) by Ehret and Powers (1957) has revealed that the hexagonal depressions correspond to regular series of cavities, the alveoli. All alveoli collectively form a continuous alveolar layer, which is delimited by an outer alveolar and inner alveolar membranes.
The outer layer lies in close contact beneath the outer cell membrane. Therefore, pellicle includes outer cell membrane, outer alveolar membrane and inner alveolar membrane.
(iii) Cilia:
The entire body is covered with numerous, small, hair like projections called cilia. Cilia occur in longitudinal rows all over the body, this condition is known as holotrichous in which the body cilia are equal. Cilia have the same structure as flagella, they have an outer protoplasmic sheath or plasma membrane with nine double longitudinal fibrils in a peripheral ring. In some cilia the nine outer fibrils are not paired.
ADVERTISEMENTS:
There are two central longitudinal fibrils which are thinner than the outer fibrils. Each cilium arises from a basal granule or kinetosome. The nine pairs of peripheral fibrils fuse together to form the wall of the kinetosome, thus, kinetosome is a tube which is either open or closed at its lower end, the two central fibrils stop at the level of the pellicle in most ciliates.
Arising from the kinetosome is a thin rhizoplast which does not join the nucleus. Many Metazoa also have cilia, their structure is the same, except that the basal granule is different and it has fine filaments or rooting fibres extending down into the cytoplasm. But cilia differ from flagella in being generally more numerous and shorter in size.
The ciliature may be conveniently divided into body or somatic cilia which are found on the body surface, and into oral ciliature which is associated with the mouth region. The body cilia are equal but they are longer at the posterior end, hence, the name caudatum. The cilia are organelles of locomotion and food collection, they also act as sensory receptors and detect the stimuli of the external environment.
Ultra Structure of Cilia:
The cilia and flagella have a fibrillar composition. At the base the cilium has the diameter of about 0.2 micron or 2,000 A0 which may be up to 10 microns above the cell surface. The cilia are bounded by a unit membrane of 90 A0 thickness which resembles and remains continuous with the plasma membrane. The bounded space of the cilium contains a watery substance known as the matrix.
In the matrix, there remain embedded eleven longitudinal fibrils or microtubules. Out of the eleven fibrils, two are located in the centre, while the remaining nine fibrils remain arranged peripherally around the central fibrils. Each of the nine outer fibrils is 360A0 in diameter and composed of two sub-fibrils of 180 to 250A0 diameter.
These sub-fibrils are designated as the sub-fibril A and sub-fibril B.
ADVERTISEMENTS:
The sub-fibril A is slightly larger than the sub-fibril B. The sub-fibril A gives out two thick projections or arms from its one side. The arms of the sub-fibril A of all the outer fibrils remain directed in clockwise direction. Further, the sub-fibril A occurs more closely to the centre of the cilium than the sub-fibril B. Both the sub-fibrils have a common wall of 50A0 thickness.
The two central fibrils do not have paired sub-fibrils like the peripheral nine fibrils but each contains only a single tubule. Each central fibril has a diameter of about 250A0 and is composed of 60A0 thick wall.
Both the central fibrils remain separated by a space of 350A0 and remain enveloped in a common sheath. Gibbnos (1967) has reported that the sheath of the central fibrils gives out nine radially oriented links or spokes to each sub-fibril A.
The high resolution electron microscopy has revealed that each of the peripheral and central fibrils of the cilia and flagella is composed of ten to twelve filaments of 40A0 thickness. Each filament is beaded. Each bead remains arranged in the lattices of 40 by 50A0 in the plane of the wall of the tubule. These beads are considered as the basic subunit of the tubule structure.
(iv) Infra-Ciliary System:
The infra-ciliary system is located just below the pellicular alveoli. It consists of the kinetosome or basal body and kinetodesma. The cilia arise from kinetosomes and from each kinetosome arises a delicate cytoplasmic fibril called kinetodesma, (Fig. 20.2).
Lying below the pellicle slightly to the right, but joined to all kinetosomes of one longitudinal row, is a longitudinal bundle of several kinetodesmata, a kinetodesmata of each kinetosome extends for a distance anteriorly into its own bundle of kinetodesmata.
A longitudinal row of kinetosomes with their kinetodesmata forms a longitudinal unit called a kinety. All the kineties or kinetia make the infra-ciliary system of a ciliate. The kinetia lie in the cortex below the pellicle, their number is fairly constant for each ciliate.
The infra-ciliary system controls and coordinates the movements of the cilia, and it brings about formation of organelles in cell division, e.g., some kinetia form the mouth. In binary fission of ciliates the kinetia are cut transversely into two, each going to one daughter cell, this is called perikinetal fission.
(v) Oral Groove and Cytopyge:
On the ventrolateral side is a large oblique, shallow depression called oral groove or peristome which gives the animal an asymmetrical appearance. It runs obliquely backwards from one side (usually left to right but in some cases right to left) and ending a little behind the middle body. The oral groove leads into a short conical funnel-shaped depression called vestibule.
ADVERTISEMENTS:
The vestibule leads directly into the fixed, oval-shaped opening called cytostome (mouth). Extending directly from the cytostome toward the centre of the body is the wide cytopharynx. The cytopharynx then turns sharply towards the posterior side to become the slender tapering oesophagus.
Thus, the oesophagus is roughly parallel to the body surface of Paramecium except at its posterior extremity. Here the oesophagus turns again toward the centre of the animal to lead into the forming food vacuole.
The cytopyge (also termed cell anus or anal spot or cytoproct) lies on the ventral surface of the body almost vertically behind the cytostome or mouth. Undigested food particles are eliminated through the cytopyge. The ciliation of cytopharynx is very complicated.
Gelei (1934) reported the presence of four rows and Lund (1941) observed at least four rows. A structure called penniculus is found on the left wall of the cytopharynx and spirals through approximately 90 degrees so that its posterior extremity is on the oral (ventral) surface of the oesophagus.
According to Lund, the penniculus consists of eight rows of cilia arranged in two closely set blocks of each. A similar band composed of four rows of long cilia which are less compact than in the penniculus is termed as quadrulus. It spirals down the dorsal wall of buccal cavity and ends close to the penniculus.
The penniculus and quadrulus have been wrongly called an undulating membrane by some workers. The quadrulus and penniculus control the passage of food. It is not known how cilia work, probably their fibrils contract in rhythmic way which causes bending. Gelei (1925) pointed out that the function of penniculus is the forcing of food elements into the body.
(vi) Cytoplasm:
The cytoplasm is differentiated into a narrow, external or cortical zone called the ectoplasm and a larger, internal or medullary region called the endoplasm.
(vii) Ectoplasm:
The ectoplasm (ectosarc or cortex) is a permanent part of the body, strikingly delimited from the endoplasm. Ectoplasm forms a firm, clear, thin and dense outer layer. It contains the trichocysts, cilia and fibrillar structures and is bounded externally by a covering called pellicle.
(viii) Trichocysts:
Embedded in the ectoplasm at right angles to the surface are small spindle-shaped bags called trichocysts. A small spot on each anterior and posterior margin of the hexagon marks the position of a trichocyst. They are filled with a refractive, dense fluid having a swelling substance, at the outer end is a conical head or spike.
The trichocysts lie perpendicular in the ectoplasm, they open by small pores on the ridges of the hexagonal areas of the pellicle.
They arise from kinetosomes of cilia, then migrate and locate themselves at equal distance in the endoplasm. When the animal is irritated, the trichocysts are discharged as long sticky threads. A discharged trichocyst has an opaque spike-like an inverted nail, and a long striated shaft, but the shaft is not seen in the un-discharged state and is probably formed during discharge.
The function of trichocysts is uncertain, but they are discharged as a reaction to local contacts and injury, they may serve as organelles of defence.
But this is uncertain because the trichocysts are ineffective against Didinium, the chief predator on Paramecium, they may be for fixing the animal to a spot during feeding. In some ciliates, the trichocysts act as organelles of offence. After trichocysts are discharged, regenerated from kinetosomes.
(ix) Neuromotorium and Associated Fibrils:
According to Lund (1933) on the left dorsal wall of the cytopharynx at about the level of the posterior margin of the cytostome is a very small, bilobed mass, the neuromotorium. From the neuromotorium, fibrils radiate into the endoplasm.
Of these four or more usually pass almost to the dorsal body wall but the rest are shorter and not definite in position. All are termed endoplasmic fibrils. Their function is unknown, but they may coordinate the feeding movements of the oral cilia. The fibrils may also give mechanical support, elasticity, contractility, conductivity and metabolic influence.
(x) Endoplasm:
The endoplasm or medulla is the more fluid and voluminous part of the cytoplasm which contains many cytoplasmic granules as well as other inclusions and structures of a specialised nature. The cytoplasmic inclusions are mitochondria, Golgi apparatuses, vacuole, crystals, granules and chromidia, etc. Other structures, viz., nuclei, contractile vacuoles and food vacuoles are also found in the endoplasm.
Nuclei:
In the endoplasm near the cytostome are two nuclei, i.e., Paramecium is heterokaryotic, a large ellipsoidal and granular macronucleus and other small compact micronucleus.
Macronucleus is a conspicuous, ellipsoidal or kidney-shaped body. It is of a compact type containing fine threads and tightly packed discrete chromatin granules of variable size and embedded in an achromatic matrix. It possesses many nucleoli and much more chromatin material (DNA).
It is somatic or vegetative nucleus. It divides amitotically and controls the vegetative functions (metabolic activities) of the animal. It does undergo mitosis.
Micronucleus is small, compact and spherical. It is generally found close to the macronucleus often in a concavity. Fine chromatin granules and threads are uniformly distributed throughout the structure. The micronucleus divides mitotically and controls the reproduction.
The number of micronucleus varies with the species; it is one in P. caudatum, two in P. aurelia and many in P. multimicronucleatum. The micronucleus contains a distinct nucleolus in P. aurelia but it is not found in P. caudatum. Moses (1949; 1950) has reported that the macronucleus and micronucleus are identical in chemical composition.
(xi) Contractile vacuoles:
There are two large, liquid-filled contractile vacuoles, each situated near one end of the body close to the dorsal surface. Their position is fixed (unlike Amoeba), they lie between the ectoplasm and endoplasm, but they are temporary organelles disappearing periodically. In some species, they appear to have a lining membrane, in which case they do not disappear entirely during systole.
Connected to each contractile vacuole are five to twelve tubular radiating canals, each consisting of a terminal part, long ampulla which collapses when empty, and a short injector canal which opens into the vacuole.
The canals communicate with a large part of the body from where they take up liquids and pour them into the vacuole which is, thus, reconstituted and grows in size, when the contractile vacuole reaches its maximum size it contracts suddenly (systole) and discharges its contents through a permanent pore in the pellicle, then the canals again form the contractile vacuoles, the canals do not disappear entirely since they are permanent structures.
The two contractile vacuoles discharge irregularly, the posterior one contracts more rapidly because it is near the cytopharynx and more water comes into it. The main function of the canals and the contractile vacuoles is hydrostatic, they remove excess of water from the protoplasm, the water is partly absorbed and partly taken in while feeding.
The nitrogenous waste substance has ammonia compounds and some urates which are expelled from the contractile vacuoles along with CO2 but there is no evidence of the excretory matter being secreted by the protoplasm into the canals or contractile vacuoles.
There is a fixed permanent cytopyge or anus below the peristome to one side of the cytopharynx, through which undigested remains of food and urates are expelled.
The electron microscopic study of contractile vacuoles has revealed that each contractile apparatus consists of some of the tubules of endoplasmic reticulum, nephridial tubules, feeder canals, accessory vacuoles (radial canals) and main contractile vacuole. The accessory vacuoles are supposed to be the ampullae of feeder canals (Fig. 20.11).
(xii) Food vacuoles:
These are roughly spherical, non-contractile bodies varying in size and number lying in the endoplasm. They contain ingested food particles, principally bacteria and a small amount of fluid bounded by a thin definite membrane. Volkonsky (1934) proposed the name gastrioles for these vacuoles. Associated with the food vacuoles are the digestive granules.
3. Locomotion of Paramecium Caudatum:
Paramecium Caudatum performs locomotion by two methods, viz., metaboly or body contortions and by cilia.
(i) Metaboly or Body Contortions:
The body of Paramecium Caudatum possesses elasticity, it can squeeze itself through a passage narrower than its body, after which the body assumes its normal shape. This temporary change of body shape is metaboly, it is brought about in Paramecium by the protoplasm.
(ii) Ciliary Locomotion:
Locomotion brought about by cilia is the main method. The cilia can beat forwards or backwards enabling the animal to swim anteriorly or posteriorly.
Normally the animal swims forwards, the cilia beating backwards but obliquely, the cilia stiffen and bend backwards rapidly to almost touch the body surface, this is called the effective stroke; then the cilia become limp and return slowly to the original vertical position, this is , called recovery stroke.
Cilia of the same transverse row beat together and those of the same longitudinal row beat one after the other from the anterior to the posterior end.
This coordinated movement of cilia is called metachronal rhythm, which is due to the infra-ciliary system; this causes swimming forward by the animal. But when the body cilia are beating obliquely backwards, then at the same time the longer cilia of the oral groove beat more vigorously which causes the anterior end to swerve to the left.
The action of cilia of body and oral groove makes the animal to rotate on its long axis. This rotation is always to the left (except in P. calkinsi which rotates in a right hand spiral).
This combination of forward motion, swerving and rotation makes the animal move forwards in a counter-clockwise spiral path. This path has a straight axis, and the same body surface of the animal remains towards the axis of the spiral path. But in swimming backwards, all species rotate to the right.
The ciliary beat can be reversed so that the cilia move obliquely forwards by which the animal swims backwards. By ciliary action, Paramecium moves with a speed of 1500 microns or even more per second.
Jennings contended that the spiralling of Paramecium is due to the fact that while the cilia strike chiefly backward they do so obliquely to the right thereby causing the animal to roll over to the left.
Also this swerving of the body toward the aboral surface is due largely to the greater power of the effective stroke of the oral cilia which strike more directly backward. The result—the rotation of Paramecium on its long axis—thereby enables the Paramecium to follow a more or less straight course in forming large spirals.
4. Nutrition of Paramecium Caudatum:
In Paramecium Caudatum, nutrition is holozoic. The food comprises chiefly bacteria and minute Protozoa. Paramecium does not wait for the food but hunts for it actively.
It is claimed that Paramecium Caudatum shows a choice in the selection of its food, but there seems to be no basis for this though it engulfs only certain types of bacteria; available data suggest that 2 to 5 million individuals of Bacillus coli are devoured by a single Paramecium in 24 hours. It also feeds on unicellular plants like algae, diatoms, etc., and small bits of animals and vegetables.
Feeding Mechanism:
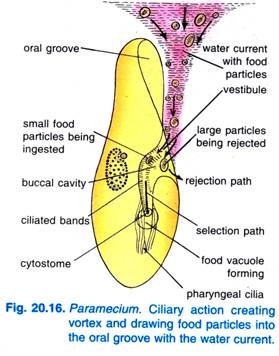
When Paramecium Caudatum enters a region of abundant food, it comes to rest. It feeds only at rest or when swimming very slowly, it never feeds when swimming fast. The beating of cilia of the oral groove causes a cone-shaped vortex of food-laden water to be swept into the oral groove from a distance in advance of the anterior end (Fig. 20.16).
The particles of food then go to the vestibule from where some food particles are rejected and thrown out, but others pass into the cytostome.
At the end of the cytopharynx, a food vacuole is formed which gets filled with particles of food. The quadrulus and peniculi control the passage of food into the food vacuole which is formed laterally. When the food vacuole reaches a certain size the post-buccal fibres clasp the food vacuole and it is pinched off by them and started on its course.
The vacuole contains some water besides the food. Rotary streaming movements of endoplasm called cyclosis carry the food vacuoles along a definite course which is functionally equivalent to a digestive tract.
The tract begins from the end of the cytopharynx, then to the posterior side, then forwards to circulate with the endoplasm, then to the dorsal surface, then towards the anterior end, then downwards to the cytopyge. Early on its journey the food vacuole decreases in size, then increases again.
Digestion and Egestion of Paramecium Caudatum:
During cyclosis, digestion occurs by enzymes secreted by protoplasm into the vacuoles. In digestion, proteins are changed into amino acids, carbohydrates into soluble sugars and glycogen, and fats are probably also digested.
The contents of food vacuoles are at first acidic (pH about 4) and then become alkaline, major digestion occurs during the alkaline phase. The undigested matter is egested through the cytopyge with some force.
Cyclosis can be demonstrated experimentally; if milk stained with Congo red is fed to Paramecium, the fat globules of milk in the food vacuoles will first turn red due to acidic reaction of enzymes, then they will change from shades of purple to blue due to alkaline reaction, the vacuoles will show the course of cyclosis.
5. Respiration and Excretion of Paramecium Caudatum:
The exchange of gases (oxygen and carbon dioxide) takes place through the semi-permeable pellicle like other freshwater protozoans by the process of diffusion. Paramecium Caudatum obtains its oxygen from the surrounding water. Carbon dioxide and organic wastes like ammonia resulting from metabolism are probably excreted by diffusing outward into the water in the reverse direction.
6. Osmoregulation in Paramecium Caudatum:
Paramecium Caudatum has two contractile vacuoles, one anterior and one posterior. The function of the contractile vacuoles is osmoregulation, i.e., to regulate the water contents of the body and may serve also in excretion of nitrogenous wastes such as urea and ammonia.
Excess of water (because of continuous endosmosis) within cytoplasm is secreted into the tubules of endoplasmic reticulum and goes to nephridial tubules → feeder canals → and collect into ampulla of a series of 6 to 11 radiating canals that converge toward and discharge into each vacuole. The canals are most conspicuous as a vacuole is forming.
When each vacuole is swelled (diastole) to a certain size, it contracts (systole) and discharges to the exterior probably through a pore. The contractile vacuoles contract alternately, at intervals of 10 – 20 seconds.
The posterior contractile vacuole works faster than anterior vacuole because of intake of large amount of water into the posterior region by the cytopharynx. The contractile vacuoles maintain an optimum concentration of water in the body cytoplasm by disposing of the excess.
7. Behaviour of Paramecium Caudatum:
The responses of Paramecium Caudatum to various kinds of stimuli are learned by study of its reactions and of the grouping or scattering of individuals in a culture. The response is positive if the animal moves toward a stimulus and negative when it moves away. To an adverse stimulus the animal continues to give the avoiding reaction until it escapes.
In avoiding reaction, the ciliary beat reverses, the animal moves backward a short distance, and then rotates in a conical path by swerving the anterior end aborally while pivoting on the posterior tip. All adjustments are made by trial and error. Experiments have shown that the anterior end of the animal is more sensitive than the other parts.
The responses of Paramecium to different stimuli may be grouped as follows:
(i) Reactions to contact (Thigmotaxis):
Response to contact is varied in Paramecium. If the anterior end is lightly touched with a fine point, a strong avoiding reaction occurs. When a swimming Paramecium collides with some object in the water, but if touched elsewhere there may be no response. A slow moving individual often responds, positively to contact with an object by coming to rest upon it.
(ii) Reactions to chemicals (Chemotaxis):
Generally Paramecia respond to a chemical stimuli by means of avoiding reaction. If a drop of weak salt solution (0.5 per cent) is introduced in a Paramecium population on a micro-slide, the animals respond with the avoiding reaction and none enters the drop. To acids, however, the response is positive even when the concentration is of sufficient strength to kill them.
(iii) Reactions to temperature (Thermotaxis):
Paramecium seeks an optimum temperature of 24 to 28°C. When a temperature change occurs markedly above or below the optimal range, Paramecia show an avoiding reaction. Greater heat stimulates rapid movement and avoiding reactions until the animals escape or are killed.
(iv) Reactions to light (Phototaxis):
With the exception of the green Paramecium bursaria, which is positively phototactic, other species are indifferent to ordinary light. However, when the light intensity is suddenly and sharply increased, a negative reaction generally follows. Paramecia exhibit an immediate negative response to ultraviolet rays.
(v) Reactions to electric current (Galvanotaxis):
Paramecia respond to electric stimuli. When two electrodes are placed opposite each other in a shallow dish containing Paramecia and a constant current applied, all the organisms swim in the same direction toward the cathode or negative electrode where they concentrate in large numbers.
If the direction of the electric current is reversed while the Paramecia are swimming toward the cathode, the organisms reverse the direction and swim toward the new cathode.
(vi) Reactions to water current (Rheotaxis):
Paramecia show a positive rheotaxis. In a gentle water current the Paramecia will mostly move with the flow with their anterior ends upstream.
(vii) Reactions to gravity (Geotaxis):
Paramecia generally exhibit a negative response to gravity as seen in a culture where many individuals gather close under the surface film with their anterior ends uppermost. If Paramecia are introduced in an inverted water filled U-shaped tube stoppered at both the ends, they immediately move upward into the horizontal part of the tube.
8. Reproduction in Paramecium Caudatam:
Paramecium Caudatum reproduces asexually by transverse binary fission and also undergoes several types of nuclear re-organisation, such as conjugation, endomixis, autogamy, cytogamy and hemixis, etc.
(i) Transverse Binary Fission:
Transverse binary fission is the commonest type of asexual reproduction in Paramecium. It is a distinctly unique asexual process in which one fully grown specimen divides into two daughter individuals without leaving a parental corpse.
The plane of division is through the centre of the cell and in a plane at right angles to the long axis of the body. Division of the cell body as a whole is always preceded by division of the nuclei; indeed it appears that reproduction is initiated by nuclear activity and division.
Paramecium Caudatum reproduces by transverse binary fission during favourable conditions. In binary fission, the micronucleus divides by mitosis into two daughter micronuclei, which move to opposite ends of the cell. The macronucleus elongates and divides transversely by amitosis.
Another cytopharynx is budded off and two new contractile vacuoles appear, one near anterior end and another near posterior end. In the meantime, a constriction furrow appears near the middle of the body and deepens until the cytoplasm is completely divided.
The resulting two “daughter” paramecia are of equal size, each containing a set of cell organelles. Of the two daughter paramecia produced, the anterior one is called proter and the posterior one is called opisthe. They grow to full size before another division occurs.
The process of binary fission requires about two hours to complete and may occur one to four times per day, yielding 2 to 16 individuals. About 600 generations are produced in a year.
The rate of multiplication depends upon external conditions of food, temperature, age of the culture, and population density; also on the internal factors of heredity and physiology. Naturally, if all the descendants of one individual were to survive and reproduce, the number of paramecia produced would soon equal to the volume of the earth.
The term clone is used to refer to all the individuals that have been produced from one individual by fission. All the members of a clone are hereditary alike.
(ii) Conjugation:
Ordinarily Paramecium Caudatum multiplies by binary fission for long periods of time, but at intervals this may be interrupted by the joining of two animals along their oral surfaces for the sexual process of conjugation.
Conjugation is defined as the temporary union of two individuals which mutually exchange micro nuclear material. It is unique type of a sexual process in which two organisms separate soon after exchange of nuclear material.
Sonneborn (1947), on the basis of mating behaviour of Paramecium Caudatum, has reported that each species of Paramecium exists in a number of varieties or syngens. Further, within each syngen there are a number of mating types usually two.
The mating types remain morphologically identical but they exhibit physiological differences. In P. aurelia, there are 14 syngens and 28 mating types, while in P. caudatum, there are 16 syngens and 32 mating types. Observations have been made that usually paramecia neither conjugate with members of their own mating type nor with the other varieties, but only with the second mating type of their own variety.
Factors Inducing Conjugation:
The factors inducing conjugation vary from species to species but some of them are given below:
1. Conjugation occurs usually under un-favourable living conditions; starvation or shortage of food and particular bacterial diet or certain chemicals are said to induce the process of conjugation in certain species of Paramecium.
2. Conjugation occurs after about 300 asexual generations of binary fission, or it alternates with binary fission at long intervals to rejuvenate the dying clone, i.e., it occurs in the individuals which must have passed through desirable number of asexual generations, said to be the period of immaturity, and then they become sexually mature to conjugate.
3. Conjugation occurs when there is a change in the physiological condition of paramecia, then it occurs between such individuals which are somewhat smaller in size (210 microns long) and they are at a stage which may be regarded as a period of unhealthy old age; the paramecia of this condition will die if not allowed to conjugate.
4. Sudden darkness in light conditions and low temperatures are said to induce the process of conjugation in some species.
5. Conjugation does not take place during night or darkness; it starts in early morning and continues till afternoon.
6. A proteinaceous substance in the cilia of mating type individuals is said to induce conjugation.
Process of Conjugation:
The process of conjugation differs in different species of Paramecium, but the undernoted account relates to the conjugation process of P. caudatum (Fig. 20.21).
In conjugation two Paramecium caudatum (referred to as pre-conjugants) of the opposite mating types of the same variety come together with their ventral surfaces and unite by their oral grooves; their cilia produce a substance on the surface of the body which causes adhesion of the two conjugating paramecia.
They stop feeding and their oral groove apparatus disappears. The pellicle and ectoplasm, at the point of contact, of both break down, and a protoplasmic bridge is formed between the two animals. Now, these individuals are called conjugants.
In this condition, the conjugating pair swim actively and simultaneously a series of nuclear changes take place in each conjugant as described below:
The macronucleus begins to disintegrate, it becomes loose in texture and forms a complex twisted skein, during the latter half of the conjugation period it will finally disappear being absorbed in the cytoplasm. The micronucleus of each conjugant divides twice, one of them being a reduction division.
Thus, four haploid daughter micronuclei are produced in each conjugant. Three of these four micronuclei degenerate in each, so that only one remains.
The remaining micronucleus of each conjugant divides mitotically into two unequal pronuclei or gametic nuclei forming a larger stationary female pro-nucleus and a smaller, active migratory male pro-nucleus.
The migratory pro-nucleus of one conjugant crosses over the protoplasmic bridge and fuses with the stationary pro-nucleus of other conjugant to form a synkaryon or conjugation nucleus in which the diploid number of chromosomes is restored and there has been an exchange of hereditary material.
The process has been compared with fertilisation in higher animals, but this is not fertilisation because no gametes are involved. The conjugants now (after about 12-48 hours) separate and are called ex-conjugants. The synkaryon of each ex-conjugant divides three times to form eight micronuclei in each ex-conjugant.
Four of the eight micronuclei enlarge and become macronuclei, and three of the other four micronuclei disappear.
The remaining micronucleus divides and at the same time the ex-conjugant divides by binary fission into two cells, each having two macronuclei and one micronucleus. The cells and their micronuclei divide a second time to form four paramecia from each ex-conjugant, so that each has one macronucleus and one micronucleus.
The new macronucleus, as also the micronucleus, have been made of new material. These new nuclei probably contain new and different potential which is reflected in the healthy individuals.
Significance of Conjugation:
A clone will die out if nuclear re-organisation does not occur, but the clone can be rejuvenated to regain its former vigour by nuclear rearrangement, this nuclear re-organisation is brought about by conjugation, thus, conjugation is essential for continued binary fission.
The significance of conjugation has been summarised below:
1. Conjugation serves as a process of rejuvenation and re-organisation by which the vitality of the race is restored. If conjugation does not occur for long periods, the paramecia weaken and die. (Woodruffs claim of keeping paramecia healthy for 22,000 generations without conjugation is disproved by Sonneborn, because he showed that all of Woodruffs paramecia belonged to the same mating type).
2. There is no distinction of sex in conjugants though only paramecia of two different mating types of the same variety will conjugate.
3. There is no distinction of sex, yet the active migratory pro-nucleus is regarded as male and the stationary pro-nucleus as the female.
4. Conjugation is only a temporary union, there is no fusion of cytoplasm and no zygote is produced, but the nucleus of each ex-conjugant contains hereditary material from two conjugating individuals.
5. Conjugation brings about replacement of the macronucleus with material from the synkaryon, this is an event of fundamental importance. In binary fission the chromosomes of the macronucleus were distributed at random to the daughter cells, continued binary fission had made the clone weak with some structural abnormalities.
Conjugation brings about the formation of the correct number of chromosomes in the macronucleus, so that the race is renewed in vigour. The role of the micronucleus is to restore a balanced chromosome and gene complex.
9. Aberrant Behaviour in Reproduction in Paramecium Caudatum:
Paramecium Caudatum shows certain variations in its nuclear behaviour during fission and conjugation, these deviations are endomixis, autogamy, cytogamy and hemixis. In the first three processes genetic recombination is effected and a new macronucleus is formed from the micronucleus.
(i) Endomixis:
Woodruff and Erdmann (1914) first of all reported a new nuclear re-organisation process, endomixis (Gr., endon = within; mixis = mingling) in Paramecium aurelia, a bimicronucleate species (Fig. 20.22). This process was described as occurring periodically in which a new macro-nuclear apparatus is produced without synkaryon formation. Endomixis occurs within a single individual.
According to Woodruff and Erdmann, the macronucleus degenerates and the micronuclei divide twice to form eight micronuclei. Six micronuclei degenerate and two remain. With only two micronuclei remaining, the animal divides by fission into two cells, each with one micronucleus.
The micronucleus of each daughter cell divides twice to form four micronuclei. Two micronuclei enlarge to form macronuclei. The animal and its micronuclei divide so that two daughter individuals are formed, each having one macronucleus and two micronuclei.
Endomixis occurs in that variety of P. aurelia which does not conjugate, hence, the effect of endomixis may be the same as that of the conjugation since both processes bring about replacement of the macronucleus with material from the micronucleus, and both processes rejuvenate the vitality of the race.
But the two processes differ because there is no fusion of pronuclei in endomixis; endomixis may be compared to parthenogenesis.
However, some workers have claimed with good reasons that endomixis is not a valid process and it has been described due to faulty observation. In all probability endomixis does not take place and it may only be a specialised case of autogamy.
Later, Erdmamm and Woodruff (1916) reported endomixis in Paramecium caudatum. Diller, however, does not believe in the validity of this process and feels that Erdmann and Woodruff have simply combined the stages of hemixis and autogamy into one scheme, endomixis.
(ii) Autogamy:
Diller (1934, 1936) and Sonneborn (1950) described a process of self-fertilization or autogamy occurring in single individual in Paramecium aurelia (Fig. 20.23). He reported that in autogamy three micro-nuclear (pregamic) divisions, involving maturation, produce the gametic nuclei (pronuclei).
During autogamy in P. aurelia, the two micronuclei divide twice (once meiotically) to form eight micronuclei, six of which degenerate. Meanwhile the macronucleus grows into skein-like mass which breaks into pieces later to be absorbed in the cytoplasm. Two of the eight micronuclei, as pronuclei, enter a protoplasmic cone bulging near the cell mouth.
The two pronuclei fuse to form synkaryon. The synkaryon divides twice to form four micronuclei. Two micronuclei become macronuclei. The Paramecium and its micronuclei divide to form two daughter individuals, each with one macronucleus and two micronuclei. This process is completed in about two days.
Autogamy brings about rejuvenation of the race.
It resembles conjugation in as much as the new macronucleus is formed by material from the micronucleus, in the new macronucleus correct number of chromosomes is restored; and also in the fact that fusion of two pronuclei occurs. But autogamy differs from conjugation because only one individual takes part in autogamy and it provides both the pronuclei, it is a kind of self fertilization.
(iii) Hemixis:
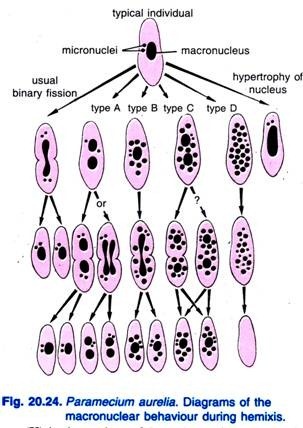
Diller (1936) reported the hemixis in Paramecium aurelia (Fig. 20.24). Hemixis is primarily a process of macro-nuclear fragmentation and division without any unusual micro-nuclear activity. Diller classified hemixis into four types, namely A, B, C, and D as shown in Fig. 20.24 in P. aurelia but he also encountered all types in mass cultures of P. caudatum and P. multimicronucleatum.
Type A is the simplest form of hemixis characterised by a division of the macronucleus into two or more parts. This division is not synchronized with micro-nuclear division.
Type B is characterised by the extrusion of one to 20 or more chromatin balls from the macronucleus into the cytoplasm.
Type C is characterised by the simultaneous splitting of the macronucleus into two or more major portions and the extrusion of macro-nuclear balls into cytoplasm.
Type D is considered to represent pathologic conditions in which the macronucleus undergoes complete fragmentation into chromatin balls that eventually disappear from the cell. Micronuclei generally disappear before the dissolution of the macronucleus.
(iv) Cytogamy:
Wichterman (1939) has reported another sexual process in Paramecium caudatum, which he termed cytogamy. In cytogamy, there is no nuclear exchange. In this process, two individuals come together by their ventral surfaces, but the pellicle of the two individuals does not break down.
The micronucleus of each individual divides thrice to form eight micronuclei, six of which disintegrate in each individual. The two remaining micronuclei fuse to- form a synkaryon in each cell. The animals now separate.
Cytogamy differs from autogamy in that there are two animals in contact with each other, but it resembles autogamy and conjugation in the fusion of two pronuclei. Cytogamy differs from conjugation in that there is no nuclear exchange between the two animals which come together.
10. Some Cytoplasmic Particles Reported in Paramecium Caudatum:
(i) Kappa Particles:
In 1938, T.M. Sonneborn reported that some races (known as killers or killer strain) of Paramecium produce a poisonous substance, called paramecin which is lethal to other individuals called sensitives. The paramecin is water soluble, diffusible and depends for its production upon some particles located in the cytoplasm of the Paramecium (killer strain).
These particles are called kappa particles. The kappa particles have DNA and RNA. A killer Paramecium may contain hundreds of kappa particles. The detailed study of these particles has revealed that a dominant gene (K) in the nucleus of Paramecium is necessary for kappa particles to exist, multiply and produce paramecin.
(ii) mµ, Particles:
R.W. Siegel (1952) reported another type of killer particles in the cytoplasm of some Paramecium. A Paramecium with mp particles is called mate killer because when it conjugates with a Paramecium without any mµ particles called mate sensitive, then it kills the latter. The mp particles are also composed of DNA, RNA, etc.
These particles exist only in those paramecia whose micronucleus contains at least one dominant gene of either of two pairs of unlinked chromosomal genes (M1 and M2).
(iii) Pi Particles:
These particles are supposed to be the mutant form of kappa particles but they do not produce any type of poisonous substance.
(iv) Lambda Particles:
These particles are reported in killer paramecia and said to produce some substance responsible for causing lysis or disintegration of sensitive paramecia, i.e., which do not possess it.