ADVERTISEMENTS:
Let us make an in-depth study of the metabolism of carbohydrates. The metabolism of carbohydrates is done through two processes: A. Catabolic Processes and B. Anabolic Processes. The catabolic processes of carbohydrates include: 1. Glycolysis 2. Citric Acid Cycle 3. Glycogenolysis 4. HMP Pathway or Pentose Phosphate Pathway and 5. Uronic Acid Pathway. The anabolic processes of carbohydrates include: 1. Glycogenesis and 2. Gluconeogenesis.
Metabolism of carbohydrates in the cell:
Metabolism is a complex process of breakdown and synthesis of the biomolecules inside the cell. Breakdown of molecules is known as catabolism and synthesis is termed as anabolism.
ADVERTISEMENTS:
A. Catabolic Process and
B. Anabolic Processes
The catabolic processes of carbohydrates includes:
(1) Glycolysis
ADVERTISEMENTS:
(2) Citric acid cycle
(3) Glycogenolysis
(4) Hexose monophosphate pathway and
(5) Uronic acid pathway.
The anabolic processes of carbohydrates include:
(1) Glycogenesis and
(2) Gluconeogenesis.
A. Catabolic Processes:
1. Glycolysis:
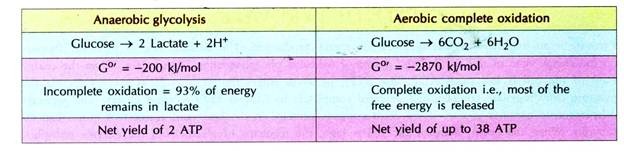
Glycolysis is the breakdown (lysis) of glucose to pyruvic acid under aerobic conditions and to lactic acid under anaerobic conditions.
Anaerobic glycolysis is also termed as Embden-Meyerhof pathway (EMP), after the scientists who proposed it. Glycolysis occurs in the cytosol of the cell and is initiated when the ATP level of the cell is low.
ADVERTISEMENTS:
It can be divided into two stages viz.:
(1) Preparatory phase and
(2) Energy yielding phase.
ADVERTISEMENTS:
In stage one, one molecule of glucose in converted into two molecules of D-glyceraldehyde-3-phosphate. Glucose is either cleaved from the glycogen molecule or enters the cell individually and is phosphorylated to glucose-6-phosphate by converting ATP to ADP with the help of the enzyme hexokinase/glucokinase.
The phosphorylation of glucose serves two purposes. First, it makes the glucose molecule more reactive and ready for other reactions. Second, because phosphorylated compounds cannot pass through the cell membrane, phosphorylation keeps the glucose inside the cell. The six carbons in glucose- 6-phosphate structure need to be rearranged to form fructose-6-phosphate so that it can split into two structures of 3 carbons each.
The new compound, fructose-6-phosphate is phosphorylated again so that each of the 2, three carbon units have a phosphate group attached to them. The conversion of fructose- 6-phosphate to fructose-6-disphosphate via phosphofructokinase is the primary regulation point of glycolysis. The final step of stage one is the splitting of fructose-6-disphosphate into 2 molecules of glyceraldehyde-3-phosphate.
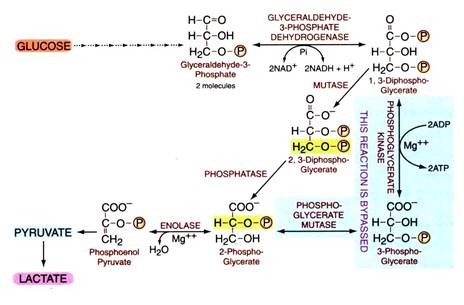
Stage 2 of glycolysis is designed to liberate inorganic phosphate for the synthesis of ATP and to convert the glyceraldehyde’s into pyruvate. Glyceraldehyde is oxidized, in other words a hydrogen atom is removed from it, and phosphorylated to produce 1,3-diphosphoglycerate.
ADVERTISEMENTS:
The NADH carries the hydrogen to the electron transfer system for the production of 3 ATPs. In the next four reactions, four additional ATPs are synthesized (two each from both the three carbon compounds), before the final product of glycolysis i.e. pyruvate is formed. The three-carbon structure of pyruvate has several fates depending upon the energy state of the cell.
 In anaerobic glycolysis, NADH + H+ is not oxidized through the electron transport chain, instead is oxidized by lactate dehydrogenase, hence no production of 6 ATPs i.e., ATPs are produced less in number.
In anaerobic glycolysis, NADH + H+ is not oxidized through the electron transport chain, instead is oxidized by lactate dehydrogenase, hence no production of 6 ATPs i.e., ATPs are produced less in number.
ATPs produced in anaerobic glycolysis = 4 (7th & 10th step)
ATPs utilized in anaerobic glycolysis = 2 (1st & 3rd step)
ADVERTISEMENTS:
Net gain of ATPs = 2 ATPs
Therefore, only two (2) ATPs are produced in anaerobic glycolysis of glucose.
Overall reaction stoichiometry/chemical summary of reaction:
i. Overall reaction:
Glucose + 2ATP + 2P, + 2ADP + 2NAD+ → 2Pyruvate + 2NADH + 2H+ + 4ATP + 2H2O
ii. Net reaction
ADVERTISEMENTS:
Glucose + 2Pi+ 2ADP + 2NAD+ 2Pyruvate + 2NADH + 2H+ + 2ATP + 2H2O
Under anaerobic conditions:
Glucose + 2 Pi + 2ADP 2 Lactate+ 2 ATP + 2H2O
(Regenerates NAD+ allowing reaction to continue in the absence of oxygen)
Anaerobic glycolysis generating lactate vs complete oxidation of glucose:
Salient features of glycolysis:
ADVERTISEMENTS:
It is the main route for glucose metabolism. It occurs in all the cells of the body. Brain and RBC depend only on glucose for oxidation and production of energy. In brain aerobic glycolysis occurs whereas in RBC there is always anaerobic glycolysis (due to the absence of mitochondria), leading to the production of lactic acid.
In skeletal muscle aerobic glycolysis occurs in normal conditions but during vigorous muscular contraction, anaerobic glycolysis is the major pathway for energy production. Although glycolysis can occur either aerobically or anaerobically, humans use aerobic glycolysis for about 90% of the time. Glycolysis can be initiated via glucose entering the cell from the blood or glucose arising from the breakdown of glycogen.
In human muscle, glycolysis is almost always initiated from the breakdown of glycogen. Since the human brain does not store glycogen, glycolysis is initiated in this tissue from blood glucose. The initiation of glycolysis is regulated by the ATP concentration in the cytoplasm. When the concentration of ATP is high and ADP is low, glycolysis is inhibited. Specifically, the enzyme phosphofructokinase is inhibited by large ATP/ADP ratio. When the concentration of ATP is low and ADP is high, glycolysis is stimulated.
Glycolysis in RBC—The Rapaport-Lumbering cycle:
Erythrocytes metabolize excessive amounts of glucose by the glycolytic pathway. This generates much ATP which is not required and cannot be used by erythrocyte.
Thus if ATP production by substrate phosphorylation is prevented by taking diversion pathway, it will:
(1) Reduce the production of ATP and
(2) Supply 2, 3-diphosphoglycerate required for the haemoglobin function which helps in de-loading of oxygen in the tissues.
Hence 1, 3-diphosphoglycerate formed in normal glycolysis is not converted to 3-phosphoglycerate, instead it takes a bypass route through 2,3-diphosphoglycerate, as under—
Fate of pyruvate:
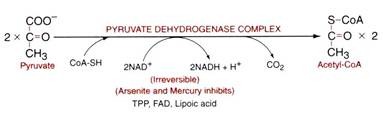
Pyruvate is an important regulatory point for energy production. The ultimate fate of pyruvate depends on the energy state of the cell and the degree of oxidative phosphorylation taking place. When the energy state of the cell is low (high ADP; low ATP), pyruvate enters the TCA cycle as acetyl-CoA via the pyruvate dehydrogenase complex and oxidized completely to CO2 & H2O to yield energy.
The pyruvate dehydrogenase complex is one of the most complex proteins in the body and consists of more than 60 subunits. When the energy state of the cell is high, the regulator of glycolysis is the enzyme phosphofructokinase, and thus there is limited pyruvate in the cell.
However, if pyruvate is present during the time of high-energy states, such as the liver metabolism of fructose, pyruvate is transformed into acetyl- CoA and is packaged as lipid. If oxygen to the cell is limiting, such as during intensive exercise, glycolysis proceeds anaerobically and pyruvate is converted to lactate by the lactate dehydrogenase enzyme. Finally, pyruvate can be converted into the amino acid alanine via transamination.
Pyruvate dehydrogenase complex is a multi-enzyme complex made up of 3 enzymes viz:
(1) Pyruvate Dehydrogenase
(2) Dihydrolipoyl Transacetylase and
(3) Dihydrolipoyl Dehydrogenase.
This reaction requires five coenzymes viz.:
(i) Thiamine pyrophosphate
(ii) Lipoic acid
(iii) Coenzyme-A (CoA)
(iv) Flavin adenine dinucleotide (FAD) and
(v) Nicotinamide adenine dinucleotide (NAD+).
Acetyl-CoA formed in the above reaction may take part either in its oxidation to carbon dioxide and water, through TCA cycle, or formation of lipids, or synthesis of cholesterol etc. etc., which depends upon the nutritional state of the body and the type of the cell where it is formed.
2. Krebs’s Cycle/Citric Acid Cycle/TCA Cycle:
Citric acid cycle also known as tricarboxylic acid (TCA) cycle is named after the scientist Sir Hans Krebs (1900-1981) who discovered it. He proposed the key elements of this pathway in 1937 and was awarded the Nobel Prize in Medicine for the discovery in 1953.
Krebs’s cycle is a set of continuous reactions (8 steps) occurring in a cyclic manner in the mitochondrial matrix in eukaryotes and within the cytoplasm in prokaryotes. Acetyl-CoA, the fuel of TCA cycle, enters the citric acid cycle inside the mitochondrial matrix, and gets oxidized to CO2 and H2O while at the same time reducing NAD to NADH and FAD to FADH2. The NADH and FADH2 can be used by the electron transport chain to create ATP.
Step 1—
Condensation:
In step 1, the two-carbon compound, acetyl-S-CoA, participates in a condensation reaction with the four-carbon compound, oxaloacetate, to produce citrate, a six carbon compound catalysed by the enzyme citrate synthase. This is the first stable tricarboxylic acid in the cycle and hence the name TCA cycle.
Step 2—
Isomerization of citrate:
Step 2 involves moving the hydroxyl group in the citrate molecule so that it can later form an α-keto acid. This process involves a sequential dehydration and hydration reaction, to form the D-isocitrate isomer (with the hydroxyl group now in the desired α-location), with cis-aconitase as the intermediate. A single enzyme, aconitase performs this two-step process.
Step 3—
Generation of CO2 by an NAD linked enzyme:
Oxidative decarboxylation takes place in the next reaction. The reaction is catalysed by the enzyme isocitrate dehydrogenase. The reaction involves dehydrogenation to oxalosuccinate, an unstable intermediate which spontaneously decarboxylates to give α-ketoglutarate. In addition to decarboxylation, this step produces a reduced nicotinamide adenine dinucleotide (NADH) co factor, or a reduced nicotinamide adenine dinucleotide phosphate (NADPH) cofactor.
Step 4—
A second oxidative decarboxylation step:
This step is performed by a multi-enzyme complex, the α-ketoglutarate dehydrogenation complex. The multi-step reaction performed by the α-ketoglutarate dehydrogenation complex is analogous to the pyruvate dehydrogenase complex, i.e. an α-keto acid undergoes oxidative decarboxylation with formation of an acyl-CoA i.e. succinyl-CoA.
Step 5—
Substrate-level phosphorylation:
Succinyl-CoA is a high potential energy molecule. The energy stored in this molecule is used to form a high energy phosphate bond in a guanine nucleotide diphosphate (GDP) molecule. Most of the GTP formed is used in the formation of ATP, by the action of nucleoside di-phosphokinase.
Step 6—
Flavin-dependent dehydrogenation:
The succinate produced by succinyl CoA-synthetase in the prior reaction needs to be converted to oxaloacetate to complete the Krebs’s cycle. The first step in the conversion is the dehydrogenation of succinate to yield fumarate facilitated by the enzyme succinate dehydrogenase. FAD is covalently bound to the enzyme (via a histidine residue) which is converted to FADH2 that is oxidized through the ETC producing 2 ATPs.
Step 7—
Hydration of a carbon-carbon double bond:
Fumarate undergoes a stereo-specific hydration of the C=C double bond, catalysed by fumarate hydratase (also known as fumarase), to produce L- malate.
Step 8—
Dehydrogenation reaction that will regenerate oxaloacetate:
L-malate (malate) is dehydrogenated to produce oxaloacetate by the enzyme malate dehydrogenase during which one molecule of NAD+ is converted to NADH 4- H+. The formation of oxaloacetate completes the Krebs’s cycle
The sum of all reactions in the citric acid cycle is
Acetyl-CoA + 2H2O + 3NAD+ + Pi + GDP + FAD à 2CO2 + 3NADH + GTP + CoASH + FADH2 + 2H+
Number of ATP’s produced in one TCA cycle:
The TCA cycle produces 3 NADH + H+ and one FADH2, these are known as the reducing equivalents. These reducing equivalents are oxidized through the electron transport chain. When NADH is oxidized through ETC it produces 3 ATPs and oxidation of FADH2 through ETC produces 2 ATPs.
Regulation of TCA cycle:
The regulation of the TCA cycle is largely determined by substrate availability and product inhibition.
i. NADH, a product of dehydrogenases in the TCA cycle, inhibits pyruvate dehydrogenase, isocitrate dehydrogenase and α-ketoglutarate dehydrogenase and also citrate synthase.
ii. Succinyl-CoA inhibits succinyl-CoA synthase and citrate synthase. ATP inhibits citrate synthase and α-ketoglutarate dehydrogenase.
iii. Calcium is used as a regulator, it activates isocitrate dehydrogenase and a-ketoglutarate dehydrogenase. This increases the reaction rate of many of the steps in the cycle, and therefore increases flux throughout the pathway.
Importance of citric acid cycle or amphibolic role of TCA cycle:
TCA cycle is the common pathway for the oxidation of carbohydrates, fats and proteins (catabolic role). The anabolic role is synthesis of various carbohydrates, amino acids and fats. As it takes part both in anabolism and catabolism, it is said to be amphibolic pathway of metabolism.
Anaplerosis:
It is the replenishment of the depleted intermediates of TCA cycle. As the TCA cycle takes part in the anabolic reactions, the intermediates of TCA cycle are utilized for the synthesis of various compounds. This results in the deficiency of one or more of the TCA cycle intermediates.
In order to continue the TCA cycle, those intermediates, which are deficient, must be filled up by some other process and this process is known as anaplerosis. For example oxaloacetate is utilized for the synthesis of the amino acid aspartic acid and oxaloacetate is replaced via anaplerosis by carboxylation of pyruvate to oxaloacetate by the enzyme pyruvate carboxykinase.
Total number of ATPs produced when glucose is completely oxidized to CO2 and H2O
(1) Glycolysis = 8 ATP
(2) 2 pyruvate 2 acetyl-CoA 2 NADH → 3×2 = 6 ATP
(3) 2 cycles of citric acid cycle for the 2 acetyl-CoA → 12×2 = 24 ATP
(4) Total = 38 ATP
(a) Total 38 ATPs are formed when one molecule of glucose is completely oxidized to CO2 and H2O.
(b) Net gain of 36 ATP is seen when NADH produced in glycolysis in the step catalysed by glyceraldehyde-3-phosphate dehydrogenase in the cytosol is transported to the mitochondria for oxidation in ETC, facilitated by glycerol phosphate shuttle instead of malate-aspartate shuttle.
(c) Net gain of 39 ATP does occur when glucose present in the glycogen is directly oxidized.
Shuttle systems:
There are some reactions that take place in the cytosol which produce NADH. These NADH have to be oxidized through the electron transport chain situated in the inner mitochondrial membrane. NADH is not permeable to the mitochondrial membrane; therefore shuttle systems operate for its transport.
There are three shuttle mechanisms:
(1) Glycerophosphate shuttle
(2) Malate- Aspartate shuttle and
(3) Isocitrate shuttle.
Glycogen metabolism:
Glycogen is a polysaccharide made up of glucose. It is the storage form of glucose in the body. Glucose requires more water for storage, but glycogen can be stored with much less amount of water hence glucose is stored as glycogen in the cell.
The largest amount of glycogen is stored in the liver and muscle. Liver glycogen provides glucose to other cells and maintains the blood glucose level in normal amounts. Muscle glycogen serves as readily available source of glucose during vigorous exercise, for glycolysis in the muscle itself. Glycogen metabolism includes glycogenesis and glycogenolysis.
3. Glycogenolysis:
Breakdown of glycogen to glucose is known as glycogenolysis.
Glycogen phosphorylase is the key enzyme of glycogenolysis. It acts only on α-1 → 4 glycosidic linkages and thus releases glucose units one by one from the linear chain, till two or three or four glucose units near the branching point are left over.
The remaining three glucose units linked by α-1 → 4 linkages are transferred to another linear chain by the enzyme glucan transferase, thus leaving one glucose residue linked with α-1 → 6 glycosidic linkage, which is acted upon by de-branching enzyme (amylo-l,6-glycosi- dase) and thus releasing free glucose. If glycogen is subjected to the action of phosphorylase alone, it will result in the formation of a glycogen molecule with each branch having only 4 glucose units which is called the ‘limit dextrin’.
Regulation of glycogen metabolism:
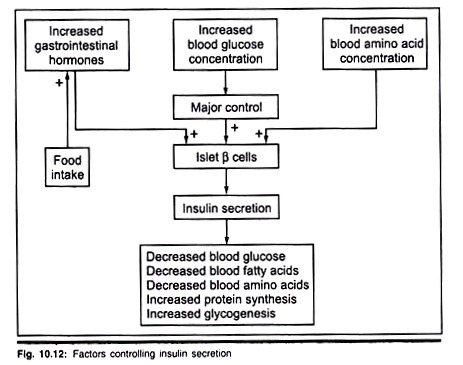
Glycogen metabolism is reciprocally regulated, mainly by the action of hormones. At the time of shock and excitement, epinephrine stimulates glycogenolysis, both in muscle and liver, whereas glucagon stimulates glycogenolysis only in the liver under hypoglycemic conditions. Insulin inhibits glycogenolysis and promotes glycogenesis.
Glycogen storage diseases:
Glycogen storage diseases are a group of inherited disorders characterized by deficient mobilization of glycogen and deposition of abnormal forms of glycogen.
4. HMP Pathway or Pentose Phosphate Pathway:
Hexose monophosphate shunt pathway or the HMP pathway is an alternative pathway for glucose oxidation. It neither utilizes nor produces ATP.
The main purpose or significance of this pathway is:
I. It produces the reducing equivalents NADPH + H+, for the synthesis of lipids (fatty acids and steroids) and keeps glutathione in reduced state in RBC.
II. It generates ribose sugar (pentose phosphate) for the formation of nucleic acids.
The organs in which HMP pathway occurs are those which are actively concerned with lipid synthesis, like the adipose tissue, kidney, lactating mammary gland, liver, RBC, thyroid and gonads. It takes place in the cytosol.
The steps involved in this pathway are:
Transketolation reaction:
Transfer of 2-carbon moiety i.e., active glycelaldehyde is known as transketolation. It is catalysed by the enzyme transketolase and the coenzyme is Thaimine pyrophosphate (TPP). In thiamine deficiency (also in pernicious anemia) transketolase activity is decreased in blood.
Transaldolation reaction:
Transfer of 3-carbon moiety i.e., active dihydroacetone is known as transaldoladon. It is catalysed by the enzyme transaldolase.
5. Uronic Acid Pathway:
This is a synthetic pathway for the various uronic acids.
Its importance is:
1. It produces glucuronic acid which takes part in detoxification of bile pigments, phenols, aromatic acids and steroid hormones.
2. It provides glucuronic acid and galacturonic acid for the formation of glycoproteins.
3. In lower animals this pathway leads to synthesis of ascorbic acids (vitamin C).
Metabolism of Fructose:
In the diet fructose is obtained from fruits, honey and table sugar (sucrose). In human body it is the sugar of the semen and amniotic fluid.
Essential fructosuria:
It is a genetic defect in which there is excretion of fructose in the urine due to the lack of the enzyme fructokinase.
Fructose intolerance:
A person shows disliking towards fruits and fructose rich diets due to the deficiency of the enzyme aldolase-B.
Metabolism of Galactose and Synthesis of Lactose:
In the diet, galactose is mainly derived from the milk sugar lactose. In the body it is converted to glycogen or may take part in the synthesis of the milk sugar lactose in lactating mammary gland.
Lactose intolerance type-II:
This is due to the deficiency of the enzyme galactose-1-phosphate uridyl transferase, which results in the accumulation of galactose in the blood i.e. galactosemia and excretion in the urine i.e. galactosuria. Such infants are intolerant to lactose and hence to milk. They show symptoms like diarrhoea and vomiting on giving milk. Lactose free milk is the only remedy.
B. Anabolic Processes:
The anabolic processes of carbohydrates are given below:
(1) Glycogenesis and
(2) Gluconeogenesis.
1. Glycogenesis:
Synthesis of glycogen from glucose is known as glycogenesis. Glucose entrapped in the cell as glucoses-phosphate is mutated to glucose-1-phosphate by the enzyme phosphoglucomutase, which in turn is attached to UTP by the enzyme glucose-1-phosphate uridyl transferase (pyrophosphorylase) forming UDP- glucose.
Glycogen synthase adds glucose (the activated UDP-glucose) to the glycogen primer (preformed glycogen with a few glucose units) by making α-1 → 4 glycosidic linkages and thus forms a linear chain of 10 to 12 glucose residues, all linked by α-1 → 4 glycosidic linkage.
At this time another enzyme i.e. branching enzyme (glycosyl-(4 → 6) transferase) removes 6 to 7 glucose units from the linear chain and transfers them to the other chain and attaches by α-1 → 6 linkage, therefore creating a branching point. The process of addition of glucose by glycogen synthase to the linear chain and branching enzyme creating the branching points is repeated and thus glycogenesis is completed.
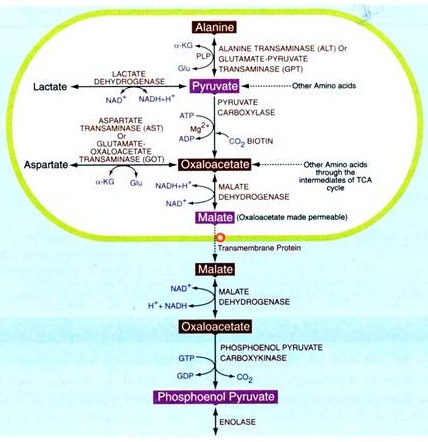
2. Gluconeogenesis:
Gluconeogenesis is the formation of glucose from non-carbohydrate sources. Gluconeogenesis helps to maintain the glucose level in the blood, so that the brain, RBC and muscle can extract glucose from it to meet their metabolic demands when dietary glucose is low. This process is very much necessary in the body because brain and RBC utilizes only glucose as energy fuel.
The major non-carbohydrate precursors of glucose are lactate, glucogenic amino acids (all except leucine) and glycerol. Lactate is formed by RBC in glycolysis because mitochondria are absent. Lactate is also formed by active skeletal muscle when the rate of glycolysis exceeds the rate of TCA cycle, the pyruvate formed is converted to lactate.
Amino acids are derived from proteins in the diet and during starvation, from the breakdown of proteins in skeletal muscle.
Glycerol is derived from the hydrolysis of triacylglycerol’s (TAG).
Gluconeogenesis occurs mainly in liver and kidney. It also occurs in brain and muscle to some extent.
Gluconeogenesis occurs during:
(1) Starvation,
(2) To clear lactate formed in RBC and muscle,
(3) When carbohydrates in the diet are low,
(4) Pregnancy,
(5) Lactation,
(6) Febrile diseases.
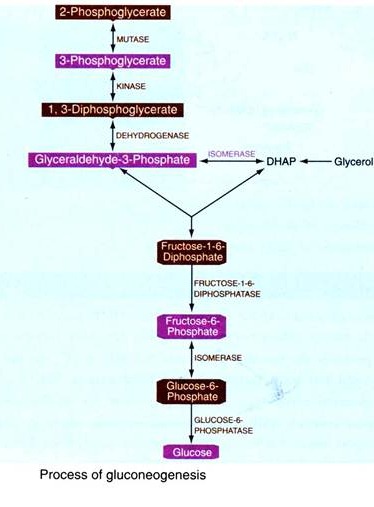
Gluconeogenesis is almost the reversal of glycolysis excepting at three steps which are irreversible in glycolysis. These steps are reversed by enzymes known as the key enzymes of gluconeogenesis i.e. those enzymes specific for gluconeogenesis only but not for any other pathway.
The key enzymes of gluconeogenesis are:
1. Pyruvate carboxylase (or carboxykinase)
2. Phosphoenol pyruvate carboxykinase
3. Fructose-1-6-diphosphatase
4. Glucose-6-phosphatase
The process of gluconeogenesis is as follows:
Role of 2, 6-Bio-phosphate in gluconeogenesis:
Fructose 2, 6-bisphosphate (or fructose 2, 6-diphosphate), is a metabolite which allosterically affects the activity of the enzymes phosphofructokinase 1 (PFK-1) and fructose 1, 6-bisphosphatase (FBPase-1) to regulate glycolysis and gluconeogenesis. Fructose 2, 6-bisphosphate is synthesized and broken down by the bi-functional enzyme, phosphofructokinase 2/fructose 2, 6-bisphosphatase (PFK-2/FBPase-2).
The synthesis of Fructose 2, 6-bisphosphate is performed through the phosphorylation of fructose 6-phosphate using ATP by the PFK-2 portion of the enzyme. The breakdown of Fructose 2, 6-bisphosphate is caused by dephosphorylation, catalyzed by FBPase-2 to produce Fructose 6-phosphate and Pi. Fructose 2, 6-bisphosphate stimulates glucose breakdown further through reduction of gluconeogenesis through allosteric inhibition of fructose 1, 6-bisphosphatase.
Hormones that regulate gluconeogenesis:
Gluconeogenesis is stimulated by:
1. Glucocorticoids
2. Thyroxine
3. Growth hormone
4. Epinephrine also stimulates but to a lesser extent
Gluconeogenesis is inhibited by:
1. Insulin
Cori’s cycle:
Conversion of muscle glycogen to liver glycogen through blood lactate and back to muscle glycogen through blood glucose is known as Cori’s cycle.
Importance of Cori’s cycle:
1. Regulation of blood sugar
2. Elimination of lactic acid from muscle