ADVERTISEMENTS:
In this article, we will discuss about the complex tissues of plant cell. Xylem and phloem are the two complex tissues which are discussed hereunder.
The complex tissues are heterogeneous in nature, being composed of different types of cell elements. The latter remain contiguous and form a structural part of the plant, adapted to carry on a specialised function.
Xylem and phloem are the complex tissues which constitute the component parts of the vascular bundle. They are also called vascular tissues.
ADVERTISEMENTS:
The vascular system occupies a unique position in the plant body, both from the point of view of prominence and physiological importance. The term ‘vascular plants’ has been in use since a long time.
In recent years a new phylum Tracheophyta has been introduced to include all vascular plants; it covers pteridophyta and spermatophyta of old classifications. Vascular bundles form a continuous and interconnected system in the different organs of the plants.
They are primarily responsible for transport of water and solutes and elaborated food matters.
Xylem:
Xylem is a complex tissue forming a part of the vascular bundle. It is primarily instrumental for conduction of water and solutes, and also for mechanical support. Primary xylem originates from the procambium of apical meristem, and secondary xylem from the vascular cambium. As a complex tissue it consists of different types of cells and elements, living and non-living.
ADVERTISEMENTS:
The tissues composing xylem are tracheids, tracheae or vessels, fibres, called xylem fibres or wood fibres, and parenchyma, referred to as xylem or wood parenchyma. Of the above mentioned elements only the parenchyma cells are living and the rest are dead. A term hadrome was once used for xylem. It included the elements excepting the fibres.
Tracheids:
A tracheid is a very much elongate cell (Fig. 538) occurring along the long axis of the organ. The cells are devoid of protoplast, and hence dead. A tracheid has a fairly large cavity or lumen without any contents and tapering blunt or chisel-like ends.
The end walls usually do not uniformly taper in all planes. Tracheids are round or polyhedral in cross-section. They are really the most primitive and fundamental cell- types in xylem from phylogenetic point of view. The wood of ancient vascular plants was exclusively made of tracheids. This is the only type of element found in the fossils of seed-plants.
In modern plants they practically occur in all groups including the angiosperms, though they predominate in lower vascular plants, the pteridophytes and gymnosperms. More effective conducting elements, tracheae or vessels, have evolved from the tracheids.
The wall is hard, moderately thick and usually lignified. Secondary walls are deposited in different manners, so that the tracheids may be annular, spiral, reticulate, scalariform or pitted. But pits of the bordered type are most abundant. Through these pits they establish communication with adjoining tracheids and also with other cells, living or non-living.
The nature of the pits on the walls of the tracheids is variable; in lower vascular plants the pits are elongated giving them scalariform appearance (Fig. 538 C & D), those of gymnosperms and angiosperms have round pits with well-developed borders (Fig. 538 A & B). Tracheids occur both in primary and secondary xylem.
Due to the presence of central lumen and hard lignified wall tracheids are nicely adapted for transport of water and solutes. They also serve as supporting tissue.
A typical fibre differs from a tracheid in more pronounced thickening of the wall and correspondingly much smaller lumen, as well as in reduction of the size of the pits. An intermediate type of cell element, called fibre-tracheid, is found in some plants.
ADVERTISEMENTS:
They have smaller pits with reduced or vestigial borders. In some cases protoplast persists up to the mature stage, and may even divide, so that transverse partition walls are noticed within the original wall. These are called septate fibre-tracheids.
These are long tube-like bodies ideally suited for the conduction of water and solutes. A trachea or vessel is formed from a row of cylindrical cells arranged in longitudinal series where the partition walls become perforated, so that the whole thing serves like a tube.
In tracheids the only openings are the pit-pairs, whereas the vessels are distinct ‘perforate’ bodies. Thus translocation of solutes becomes more easy in a vessel, as it proceeds more or less in a straight line; but the line of conduction is rather indirect in a group of tracheids.
Perforations are commonly confined to the end-walls, but they may occur on the lateral walls as well.
ADVERTISEMENTS:
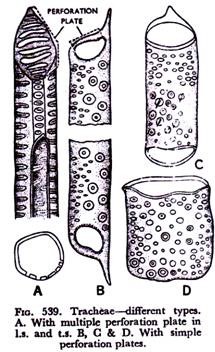
The walls undergoing perforations are referred to as perforation plates, which are mainly of two types multiple plates and simple ones. In primitive plants it has been found that the end-walls between the cells do not completely dissolve, but the openings or perforations remain either in more or less parallel series like bars called scalariform perforation (Fig. 539A) or in form of a network known as reticulate perforation, or even may form a group of circular holes (foraminate perforation).
In advanced types of plants the dissolution of the end-wall is more or less complete, and the perforation occurs in form of a single large circle. This is referred to as simple perforation (Fig. 539B).
There is anatomical evidence in support of the fact that the single large circular or oval perforation has been formed by gradual disappearance of the transverse bars of scalariform and other types. The vessels are considerably long bodies; in ash plant, Fraxinus excelsior of family Oleaceae vessels has been reported to be as long as 10 ft.
Like tracheids these elements are devoid of protoplast and have hard and lignified cell-wall with different types of localised thickenings. Some forms intermediate between typical tracheids and vessels have been noticed. These elements, analogous to fibre-tracheids, are called vessel-tracheids.
Ontogeny of a Vessel:
A vessel or a trachea originates from a row of meristematic cells of procambium or vascular cambium which remain attached end on end in longitudinal series (Fig. 540).
As usual the cells grow and secondary walls are laid down, only the primary walls where perforations will take place remain uncovered. The secondary walls undergo lignification and other changes.
The protoplast in the mean time becomes progressively more and more vacuolated and ultimately dies and disappears. The primary walls swell due to increase of pectic intercellular substance and break down, thus forming the continuous vessel.
It should be noted that a vessel or trachea arises from a group of cells, unlike a tracheid, which is an elongate ‘imperforate’ single cell. The individual cells taking part in the formation of the vessel are called vessel elements.
ADVERTISEMENTS:
The walls of the vessels are thick, hard and lignified. The secondary walls are deposited in different patterns, so that the thickenings may be ring-like, spiral, scalariform, reticulate or pitted. The pits are mostly of bordered types.
Tracheids are more primitive than the vessels. In fact, in the primitive types of vessels the form of a tracheid is maintained, but with advance in evolutionary line the diameter of a vessel may so much increase that it may become drum-shaped (Fig. 539 C & D) in appearance.
Vessels have originated phylogenetically from the tracheids; and occur in the pteridophytes Pteridium and Selaginella, in the highest gymnosperms, Gnetales, and in the dicotyledons and monocotyledons.
Comparative-studies on the dicotyledons have revealed that evolution of vessel members have proceeded from the long narrow elements with tapering ends to short ones with wider cavities having transverse or inclined end-walls which ultimately dissolved.
Suggestions about independent development of vessels by parallel evolution has also been put forward (Cheadle, 1953). Vessels first appeared in the secondary xylem and then proceeded towards primary xylem.
In some dicotyledons belonging to the families Winteraceae, Trochodendraceae and Tetracentraceae and others of the lowest taxonomic group, curiously the vessels are absent (Bailey and others).
ADVERTISEMENTS:
They do not occur in some xerophytes, parasites and aquatic plants. These have been interpreted as cases of reduction of xylem tissues involving evolutionary loss. In monocotyledons vessels are not present in secondary xylem (which tissue is lacking in many monocotyledons). Here vessels first appeared in the roots and then extended to the aerial organs (Cheadle, ’53; Fann. ’54).
These are the most important elements of xylem. They are primarily adapted for easy transport of water and solutes, and, secondarily, for mechanical support.
Xylem Fibres:
Some fibres remain associated with other elements in the complex tissue, xylem, and they mainly give mechanical support. As previously stated, fibres are very much elongated, usually dead cells with lignified walls.
Xylem fibres or wood fibres are mainly of two types: fibre-traeheids (Fig. 536 D & E) and libiriform fibres (Fig. 536 A & B) which usually intergrade, so much so that it is difficult to draw a line of demarcation between them.
Fibre-tracheids, as already reported, are intermediate forms between typical fibres and tracheids; they possess bordered pits, though the borders are not well-developed. Libiriform fibres ate narrow ones with highly thickened secondary wall.
ADVERTISEMENTS:
The central lumen is almost obliterated and pits are simple. They resemble the phloem fibres, and hence the name. They occur abundantly in many woody dicotyledons.
Phylogeny of Tracheary Elements:
The tracheary elements have developed during the evolution of land plants (Bailey, ’53). In the lower vascular plants the function of conduction and support were combined in the tracheids.
With increasing specialisation woods evolved with conducting elements—the vessel members being more efficient in conduction that in providing mechanical support. On the other hand fibres evolved as principal supporting tissue.
Thus from the primitive tracheids two lines of specialisation diverged—one toward the vessel and the other toward the fibre.
The following structural features may be taken as the basis in support of the evolution of the tracheary elements from primitive tracheids which are usually long imperforate cells with small diameter, angular in cross-section, having lignified scalariformly pitted walls.
(i) The primitive vessels are also elongate bodies like the tracheids with rather small diameter and tapering ends. Similar condition is still noticed in lower dicotyledons. With evolutionary advance they gradually become shorter and wider, often becoming drum-shaped in appearance.
(ii) The wall of the primitive tracheid is rather thin, more or less of equal thickness, and it is angular in cross-section. Same condition prevails in primitive vessels. With progressive advance considerable thickening appeared and the vessels became circular or nearly so in cross-section.
(iii) In the primitive vessels the perforation plates are multiple, usually scalariform with numerous bars, and oblique end-walls. Progressive increase in specialisation led to gradual decrease in the number of bars and their ultimate disappearance, so that the perforation plates become simple with transverse end-walls. These are positively advanced characters.
(iv) The pitting of the vessel wall also changed from early scalariform arrangement, characteristic of tracheids, to small bordered pit pairs, first in opposite (arranged in transverse rows) and ultimately in alternate (arranged spirally or irregularly) pattern. Moreover the pit pairs between vessels and parenchyma changed from bordered to half-bordered and then to simple.
In the specialisation of the xylem fibres adapted for more efficient support there has been steady increase in thickness of the wall leading to decrease in cell-lumen. The pits changed from elongate to circular, the borders becoming reduced and functionless, and ultimately disappeared. Thus the evolutionary sequence was from tracheids, through fibre-tracheids to libiriform fibres.
Xylem Parenchyma:
Living parenchyma is a constituent of xylem of most plants. In primary xylem they remain associated with other elements and derive their origin from the same meristem. In secondary xylem parenchyma occurs in two forms: xylem parenchyma (Fig. 541 A) is somewhat elongate cells and lie in vertical series attached end on end; ray parenchyma (Fig. 541 B) cells occur in radial transverse series in many woody plants.
Parenchyma is abundant in the secondary xylem of most of the plants, excepting a few conifers like Pinus, Taxus and Araucaria. These are the only living cells in xylem.
The cells may be thin-walled or thick-walled. If lignified secondary wall is present, the pit-pairs between the cells and the adjacent xylem element may be bordered, half-bordered or simple. Between two parenchyma cells the pit is obviously simple.
These cells are particularly meant for storage of starch and fatty food; other matters like tannins, crystals, etc., may also be present. As a constituent part of xylem they are possibly involved in conduction of water and solutes and mechanical support.
Phloem:
The other specialised complex tissue forming a part of the vascular bundle is phloem It is composed of sieve elements, companion cells, parenchyma and some fibres. Sclerotic cells may also be present.
Phloem is chiefly instrumental for translocation of organic solutes—the elaborated food materials in solution. The elements of phloem originate from the procambium of apical meristem or the vascular cambium.
Two terms, bast and leptome, have been used for phloem, though they are not exactly synonymous with it. Bast, derived from the word ‘bind’, was introduced before the
discovery of sieve elements; it mainly meant the fibres. The soft-walled parts of phloem, obviously excluding the fibres, were referred to as leptome.
Sieve Elements:
The most important constituents of phloem are the sieve elements, the sieve tubes and sieve cells. From ontogenetic point of view a sieve tube resembles a vessel and a sieve cell a tracheid.
Sieve tubes (Fig. 542) are long tube-like bodies formed from a row of cells arranged in longitudinal series where the end-walls are perforated in a sieve-like manner. The perforated end-walls are called the sieve plates, through which cytoplasmic connections are established between adjacent cells.
The perforations or sieve areas, as they are called, may be compared to the pit fields of the primary wall with plasmodesmata connections. But the sieve areas are more prominent than pit fields and the connecting strands are more wide and conspicuous.
It may be that a number of plasmodesmata fuse to form a connecting strand. Moreover, an insoluble substance, called callose, probably a carbohydrate of unknown chemical composition, is impregnated into cellulose or replaces cellulose forming a case round each connecting strand which passes through the sieve area (Fig. 543A).
A sieve area in surface view looks like a depression on the wall having a pretty good number of dots. Each dot represents a connecting strand in cross-section and remains surrounded by a case of callose (Fig. 543).
In sectional view sieve areas appear like thin places on the wall through which the connecting strands pass from one cell to another (Fig. 543). The sieve plate or the perforated end-wall is really the primary walls of two cells with the middle lamella in between them. The end-walls may be obliquely inclined or transverse.
A sieve plate is called simple (Figs. 542 & 543), if it has only one sieve area, whereas the plate may be compound (Fig. 544) with several sieve areas arranged in scalariform, reticulate or other manners. Though rare, the sieve areas may occur on the side walls as well. From evolutionary point of view simple sieve plates on transverse end-walls are more advanced characters than compound plates on oblique walls.
The cylindrical cells which take part in the formation of the sieve tube are called sieve tube elements. Like vessel elements the sieve tubes have also undergone decrease in length with evolutionary advance.
Sieve cells (Fig. 542C), which may be compared to the tracheids, are narrow elongated cells without conspicuous sieve areas. They usually have greatly inclined walls, which overlap in the tissue, sieve areas being more numerous in the ends.
Sieve cells are more primitive than the sieve tubes. They occur in lower vascular plants and
gymnosperms. In fact, sieve tubes have evolved from the sieve cells, as vessels have evolved from the tracheids, and so sieve tubes occur in all angiosperms. In monocotyledons, unlike the xylem elements, sieve tubes first appeared in the aerial organs, the course being from the leaves to the stem and, lastly, to the roots.
Ontogeny of the Sieve Elements:
In spite of close ontogenetic resemblance between tracheary elements of xylem and sieve elements of phloem, the latter unlike the former, are living. They originate from the mother cells (Fig 545) which are usually short cylindrical or elongate ones.
The mother cell divides longitudinally into two daughter cells, one of which serves as the sieve element and the other one becomes the companion cell, of course in those cases where companion, cells occur. The sieve element undergoes gradual differentiation. It grows in length, cytoplasm gets more and more vacuolated, so that it may have a lining layer of cytoplasm round a large central vacuole.
The most outstanding character is the disintegration of the nucleus with the maturity of the sieve elements. In fact, a distinct nucleus is present in every cell at the meristematic stage. During differentiation the nucleus disorganises (Fig. 545F).
It is the only living functioning element without a nucleus. Small colourless plastids are also present in the protoplast. They contain carbohydrates which give wine-red reaction with iodine and are interpreted as starch grains. Slimy proteinaceous bodies abundantly occur in the sieve tubes, what is commonly called slime. It is said that slime originates in the cytoplasm as small discrete bodies, which eventually fuse and get dispersed in the vacuoles.
In fixed preparations funnel-shaped slime bodies may be distinctly seen in form of plates referred to as slime plugs (Fig. 545H), on the sieve plates. In this connection a very interesting statement has come from a well-known authority, Prof. K. Esau, to the effect that in some plants the nucleolus is extruded from the nucleus before it finally disorganises and that the nucleolus persists in the tube.
Slime bodies have not been observed in pteridophytes, gymnosperms and monocotyledons. Sieve areas develop from the primary pit fields and the connecting strands originating from one or a group of plasmodesmata become more conspicuous which remain surrounded by callose cylinders.
Another theory demands that pores are formed by dissolution of cell wall and no plasmodesmata occur at the pore sites. The connecting strands were thought to be entirely cytoplasmic in nature; but it is argued that may contain vacuolar substances and thus establish connections between vacuoles of neighbouring elements.
The wall of sieve elements is primary and chiefly composed of cellulose. Thick walls are found only in exceptional cases. The tubes often cannot withstand the pressure from adjoining cells and ultimately get crushed.
It has been stated that protoplasmic strands pass through the pores of the sieve areas and that the strands remain surrounded by callose. With the differentiation of the tube the amount of callose increases and finally forms something like a pad on the sieve plate.
This pad is referred to as callus pad. Due to its formation the cell to cell communication is considerably cut down or entirely prevented. The callus pad is usually formed with the approach of resting or inactive season; and it disappears when the active season (spring) sets in. This type is Known as seasonal or dormancy callus.
In old functionless sieve tubes callus becomes permanent, what is called definitive callus. Though the term definitive callus is often used to designate the former type, it is desirable to confine it to permanent callus of old and functionless tubes.
Companion Cells:
Companion cells (Figs. 542 & 545) remain associated with the sieve tubes of angiosperms, both ontogenetically and physiologically. These are smaller elongate cells, having dense cytoplasm and prominent nuclei. Starch grains are never present.
They occur along the lateral walls of the sieve tubes. A companion cell may be equal in length to the accompanying sieve tube element or the mother cell may be divided transversely forming a series of companion cells (Fig. 545).
A sieve tube element and a companion cell originate from the same mother cell. Their functional association is evident from the fact that companion cells continue so long the sieve tubes function, and die when the tubes are disorganised.
The companion cells are so firmly attached to the sieve tubes that they cannot be normally separated by maceration. In transverse section it appears as a small triangular, rectangular or polyhedral cell with dense protoplast (Figs. 542 & 545).
In pteridophytes and gymnosperms some small parenchymatous cells remain associated with sieve cells, which are known as albuminous cells. They die in natural course when the sieve cells become functionless. Thus the relation between sieve Cells and albuminous Cells is similar to that existing between sieve tubes and companion cells, excepting that they have no common origin.
Companion cells occur abundantly in angiosperms, particularly in the monocotyledons. They are absent in some primitive dicotyledons and also in the primary phloem of some angiosperms. The wall between the sieve tube and companion cell is thin and provided with primary pit fields.
Parenchyma:
Besides companion cells and albuminous cells, a good number of parenchyma cells remain associated with sieve elements. These are living cells with cellulose walls having primary pit fields. They are mainly concerned with storage of organic food matters. Tannins, crystals and other materials may also be present.
The parenchyma cells of primary phloem are somewhat elongate and occur with the sieve elements along the long axis (Fig. 542). In secondary phloem they may be of two types.
Those which occur in vertical series are called phloem parenchyma; and others occurring in horizontal planes are known as ray cells, the position being just like the parenchyma and ray cells of secondary xylem. The cell wall is primary, composed of cellulose. Parenchyma is absent in the phloem of monocotyledons.
Fibres:
Sclerenchymatous fibres constitute a part of phloem in a large number of seed plants, though they are rare in pteridophytes and some spermatophytes. They occur both in primary and secondary phloem. These are typical elongated cells having interlocked ends, lignified walls with simple pits. The fibres of primary phloem are essentially similar to those occurring in cortex and secondary phloem.
They are of considerable commercial importance, as these fibres are abundantly used for the manufacture of ropes and cords. The flax fibres, unlike others, have non-lignified walls. Sclerotic cells are often present in primary phloem. They probably develop from parenchyma with the age of the tissue. So it is a case of ‘secondary sclerosis’.