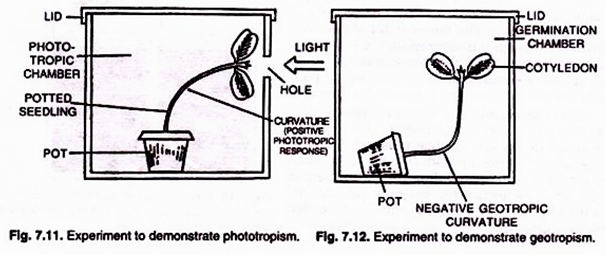
ADVERTISEMENTS:
Antibodies: Characteristics and Functions of Immunoglobulin’s (Igs) or Antibodies!
Definition:
Antibodies are immunoglobulin’s (Igs) which are produced in the body in response to the antigen or foreign bodies.
ADVERTISEMENTS:
Thus all antibodies are immunoglobulin’s but all immunoglobulin’s are not antibodies.
Location and Formation:
The antibodies may be bound to a cell membrane or they may remain free. Antibodies are produced by В lymphocytes and plasma cells. In fact B-lymphocytes get transformed into plasma cells. The mature plasma cell produces antibodies at an extremely rapid rate— about 2000 molecules per second. Antibodies direct the antibody- mediated immunity (= humoral immunity).
Types of Antibodies:
There are five types of antibodies viz:
ADVERTISEMENTS:
1. IgA (Ig alpha);
2. IgD (Ig delta);
3. IgE (Ig epsilon);
4. IgG (Ig gamma) and
5. IgM (Ig mu).
Among the antibodies, IgG forms 80% of the antibodies in the body.
Antibody Structure:
IgG has been studied extensively and serves as a model of basic structural unit of all Igs.
An antibody molecule consists of the following parts.
(i) Heavy and Light Chains:
ADVERTISEMENTS:
An antibody molecule is made up of 4 peptide chains, two small called light chains and two longer called heavy chains. Hence an antibody is represented as H2L2. The heavy chain has larger number of amino acids while light chain has smaller number of amino acids. Heavy and light chains may be either lambda or Kappa type.
(ii) Constant and Variable Regions:
There are two different regions the constant region and variable region in each chain of the antibody.
(iii) Disulfide Bonds and Hinge Region:
ADVERTISEMENTS:
A disulfide bond joins a light chain with a heavy chain. Two disulfide bonds also link the two heavy chains. This part of the antibody displays considerable flexibility and is called the hinge region. Because the antibody “arms” can move somewhat as the hinge region bends, an antibody can assume a Y shaped molecule.
(iv) Fragment Antigen Binding (Fab) and Fragment Crystallisable (Fc):
Two identical fragments of Y-shaped molecule possess the antigen-binding sites and are thus named fragment-antigen binding (Fab). The antigen-binding sites bind to the specific antigens in a lock and key pattern, forming an antigen-antibody complex. The third fragment which lacks the ability to bind to antigen and can be crystallized, is, therefore, known as fragment crystallizable (Fc).
The stem of the Y-shaped antibody monomer is called the Fc region, so named because when antibody structure was first being identified, it was a fragment (F) that crystallized (c) in cold storage.
Characteristics and Functions of Immunoglobulin’s (Igs) or Antibodies:
Antibodies show the following characteristics and perform different functions.
ADVERTISEMENTS:
(i) IgA:
It is the second most abundant class, constituting about 10 to 15 per cent of antibodies of serum. It is mainly found in sweat, tears, saliva, mucus, colostrum (first milk secreted by a mother) and gastrointestinal secretions.
Smaller quantities are present in blood and lymph. IgA has an extra polypeptide called a J-(joining) chain and extra protein known as secretory component. Levels decrease during stress, lowering resistance to infection. Provides localized protection in external secretions (tears, intestinal secretions, etc.) against bacteria and viruses. When IgA is excreted through faeces, it is called coproantibody.
ADVERTISEMENTS:
(ii) IgD:
It is mainly found on the surfaces of В cells as antigen receptors, where it activates В cells for antigen recognition. It is about 0.2% of all antibodies in the blood.
(iii) IgE:
It is less than 0.1% of all antibodies in the blood; located on mast cells and basophils releasing histamine from mast cells and basophils. It is involved in allergic and hypersensitivity reactions; provides protection against parasitic worms. This immunoglobulin was discovered in 1966 by Ishizaka.
It exhibits unique properties such as heat lability (inactivated at 56°C in one hour). IgE mediates type I hypersensitivity (anaphylaxis). Prausnitz and Kustner in 1921 demonstrated transmission of IgE-mediated type I hypersensitivity. It is called Prausnitz-Kustner (PK) reaction. Thus IgE acts as mediator in allergic response.
(iv) IgG:
ADVERTISEMENTS:
This is the most abundant class of Ig in the body constituting approximately 80% of the total Igs. It is found in the blood, lymph and intestine. It protects against bacteria and viruses by enhancing phagocytosis, neutralizing toxins and complement activation. It is the only class of antibody to cross the placenta from mother to foetus thereby conferring considerable immune protection in new-borns.
(v) IgM:
IgM is about 5 to 10% of all antibodies in the blood. It is also found in lymph. It is the largest Ig which is secreted first by the plasma cells. It is so named because it is a macroglobulin at least five times larger than IgG. IgM is the oldest immunoglobulin class. It activates the В cells. It is also the earliest immunoglobin to be synthesised by the foetus, IgM has a J chain and its each dimer contains polypeptide called a secretory component.
It cannot cross the placental barrier. IgM is 500-1000 times more effective than IgG in opsonisation (to be described ahead), in bacterial action and in bacterial agglutination. But in neutralization of toxins and viruses, it is less active than IgG. It helps in complement activation.