ADVERTISEMENTS:
Here is an essay on the ‘Life Cycle of Cycas’ for class 11 and 12. Find paragraphs, long and short essays on the ‘Life Cycle of Cycas’ especially written for school and college students.
Essay # 1. Sporophyte of Cycas:
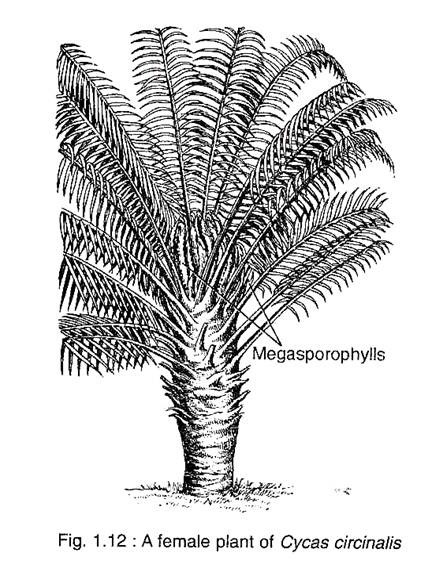
The Cycas plant looks like a palm tree with a columnar aerial trunk and a crown of pinnately compound leaves (Fig. 1.12). Generally the stem is unbranched, but older trees sometimes exhibit branching. The branch is developed from the adventitious bud called ‘bulbil’ which arises from the leaf base. Thus the bulbil helps in vegetative propagation of the plant. The stem is covered by an armour of alternating bands of large and small rhomboidal leaf bases.
These leaf bases are persistent of which the large bases belong to foliage leaves, while the small ones belong either to scale leaves (in male plants) or to sporophyll (in female plants). Cycas exhibits leaf dimorphism in possessing large green foliage leaves and small scale leaves or cataphylls. The Primary tap root system is replaced by a number of” strong, branched adventitious roots arising from the stem.
Essay # 2. Reproduction of Cycas:
Cycas reproduces both by vegetative and sexual methods:
I. Vegetative Reproduction:
Cycas reproduces vegetatively by means of bulbils or adventitious buds. These bulbils develop from the parenchymatous cells of the cortex in the base of the stem at the crevices between persistent leaf bases. Initially bulbils are covered only with scale leaves, but a few foliage leaves develop with further growth.
ADVERTISEMENTS:
Bulbil arising on male plant gives rise to male plant, if it forms on female plant it produces a female plant. This is a very common method of vegetative propagation in C. revoluta. The vegetative propagation in C. circinalis takes place by suckers that develop on roots. With further growth they start producing new plants.
II. Sexual Reproduction:
Cycas is strickly a dioecious plant, but the male and female plants are indistinguishable at the vegetative stage. The compact male cone develops at the apex of the stem in the male plant. However, Cycas is the only genus of Cycadaceae which does not produce any female cone. Instead, several megasporophylls arise spirally in acropetal succession around the stem apex of the female plant.
Male Cone:
The surface of male cone is covered with brown scales at young stage. At maturity, the male cone becomes very large (40-80 cm in length), oval or conical in shape (Fig. 1.17A) which emits odour that can be smelt from quite a moderate distance. The male cone of Cycas is the largest among the plant kingdom. The male cone has a central cone axis and numerous microsporophylls are arranged spirally and acropetally around the axis.
A single micro- sporophyll is a nearly triangular flattened woody structure. It is differentiated into a proximal wedge-shaped fertile part and a distal sterile part, tapering into an upcurved apex called apophysis (Fig. 1.17C). Numerous microsporangia (700 in C. circinalis, 1100 in C. media) are borne on the abaxial (lower) surface of the microsporophyll except at the apex and the base (Fig. 1.17B).
Microsporangia are borne in groups of 3-5 forming sori that are surrounded by many single- called delicate hairs (Fig. 1.17D). Each sporangium is oval or circular in shape having a very short massive stalk.
The dehiscence of sporangia takes place by longitudinal slit (Fig. 1.17D). The development of sporangia is of eusporangiate type. The sporangial wall is multilayered with a thickened epidermis and a ill- defined tapetum enclosing numerous microspore (pollen) mother cells.
Further, microspore mother cells through meiotic division produce numerous microspores or pollen grains. The pollen grain is oval-shaped having a large rounded monosulcate aperture. The pollen is bounded by two concentric wall layers; the outer thick exine and the inner thin intine (Fig. 1.20A).
Female Fructification:
The megasporophylls of Cycas are not organised into a definite cone. Instead, they arise at the stem apex spirally and acropetaily forming a loose crown. The megasporophylls are pinnate in nature and are covered with brown hairs called rementa.
Hence the megasporophylls of Cycas are considered to be the modified foliage leaves. Megasporophylls are produced every year like the foliage leaves, though they are produced more in numbers than the foliage leaves.
A single megasporophyll is a flat dorsiventral structure, measuring up to 30 cm in length. It is differentiated into an upper pinnate lamina and a basal stalk that bears two rows of opposite or sub-opposite, one to six pairs of ovules (Fig. 1.18A-F). There is a great variation in structure of megasporophylls and in number of ovules per megasporophyll in Cycas and these criteria can be applied to identify the species of Cycas.
ADVERTISEMENTS:
There is a great reduction in the structure of megasporophylls and in number of ovules among the various species of Cycas. C. revoluta is the most primitive species where the megasporophyll lamina is much dissected and tapers into a point, bearing 3-4 pairs of ovules (Fig. 1.18A). In C. pectinata, the mere serrated lamina is orbiculate with pectinate margins, bearing 2-3 pairs of ovules (Fig. 1.18 B).
In C. circinalis, the lamina is lanceolate with dentate margins and acuminate apex, bearing 4-6 pairs of ovules (Fig. 1.18C). The further reduction in lamina has been observed in C. rumphii (Fig. 1.18D) and C. beddomei (Fig. 1.18E) where lamina is ovate-lanceolate with acuminate apex, bearing 2-3 pairs of ovules. The maximum reduction in megasporophyll has been noted in C. normanbyana. Its lamina shows a mere serrated margins bearing only one pair of ovule (Fig. 1.18F).
Ovule:
ADVERTISEMENTS:
The ovules of Cycas are orthotropous and shortly stalked. The ovules are large, attaining a length of about 6-7 cm with a diameter of 4 cm and is perhaps the largest amongst the plant kingdom. The mature seeds are elliptical, bilobed and slightly flattened — they become fleshy and bright orange or red in colour.
There is a single integument which is fused with the nucellus except the top (Fig. 1.19). The integument is differentiated into three layers. The outer layer is fleshy and pulpy and becomes variously coloured at maturity. The middle layer is very hard and stony, while the inner layer is fleshy and becomes papery before maturity. There are frequent mucilage canals and tannin cells in the integument.
The ovule is supplied with three vascular traces (Fig. 1.19). The median strand supplies the base of the integument, which extends up to the chalazal end of the nucellus and further ramifies abruptly. The two side strands pass to the integument which again divide — one branch supplies to the outer pulpy layer and the other to the inner soft layer.
Megasporogenesis:
A deeply situated cell of the nucellus is differentiated into a large megaspore mother cell which undergoes meiotic division to form a linear tetrad of four megaspores. Out of the four megaspores the outer three degenerate, while the lowermost megaspore becomes functional (Fig. 1.21 A).
The upper free opening of the integument forms the micropyle and a concavity in the top of the nucellar tissue forms the pollen chamber. After pollination, the pollen grains are collected in the pollen chamber and the development of pollen grains takes place in the nucellar tissue.
Essay # 3. Gametophyte of Cycas:
The spore is the first phase of gametophyte generation. The microspore or pollen grain is the male gametophyte, while the megaspore represents the first stage of female gametophyte which develops to form a female gametophyte.
Development of Male Gametophyte before Pollination:
The pollen nucleus undergoes mitosis to form endosporically a small lens-shaped prothallial cell and a large antheridial initial (Fig. 1.20B). The prothalial cell does not divide further, while the antheridial initial divides into an antheridial cell and a tube cell (Fig. 1.20C). The pollen grains are released from the sporangium at the 3-celled stage. After pollination, the further development takes place within the nucellus tissue of the ovule.
Development of Male Gametophyte after Pollination:
The further development of male gametophyte takes place within a week after pollination. The tube cell of 3-celled male gametophyte comes out through the pollen aperture in the form of a pollen tube which penetrates into the nucellar tissue. The pollen tube destroys all the tissues between pollen chamber and the female gametophyte.
Thus, the fertilisation is siphonogamous, though it is haustorial in nature. Now, the antheridial cell within the pollen tube divides into a stalk cell and a spermatogenous (body) cell (Fig. 1.20D). The spermatogenous cell enlarges considerably and two blepharoplasts develop at the opposite end of the spermatogenous cell.
Then the spermatogenous cell divides to form two large, top-shaped, motile sperms with spiral band bearing numerous flagella around the distal end of the sperm (Fig. 1.20E). The sperms of Cycas are very large (180-210 pm) and can be visible to the naked eye (Fig. 1.20F).
Development of Female Gametophyte:
The female gametaphyte of Cycas develops from the functional megaspore surrounded by a tough membrane that persists in the gametophyte (Fig. 1.21B). The nucleus of the megaspore divides by free nuclear divisions forming a large number of nuclei unaccompanied by wall-formation. In the meantime, the megaspore increases in size and develops a vacuole in the center which forces the cytoplasm along with nuclei towards the periphery.
ADVERTISEMENTS:
Thus the nuclei lie in a thin film of cytoplasm around the vacuole (Fig. 1.21B). The cell wall formation starts in a centripetal fashion, from periphery inwards. The wall formation proceeds very rapidly and as a result the central vacuole in obliterated (Fig. 1.21C).
The entire gametophyte becomes cellular and the tissue thus formed is called endosperm. The peripheral cells of the gametophyte are small, isodiametric devoid of any food reserves, while the inner cells are large, irregular and filled up with starch grains.
Development of Archegonia:
Two to eight cells of the female gametophyte at the micropylar end enlarge in size and have dense cytoplasm and prominent nuclei. These cells function as archegonial initials (Fig. 1.21 D). Each archegonial initial divides periclinally to form an outer small neck initial and a large central cell (Fig. 1.21 E). The neck initial divides by a vertical (anticlinal) wall to form two neck cells (Fig. 1.21F).
A second division takes place prior to fertilisation, thus the neck actually comprises of four cells. The central cell enlarges considerably in size and its nucleus divides into a small ephemeral ventral canal nucleus and a large egg cell (Fig. 1.20G).
The egg of Cycas is reported to be the largest among the living plants, thus it can be seen with naked eyes. The nucellar tissue above the archegonia disorganises to form an archegonial chamber (Fig. 1.21G).
Pollination:
Cycas is anemophilous i.e., wind pollinated. The airborne pollen grains remain suspended in the air and at the same time some cells of the nucellar beak in the ovule are disorganised to form a viscous fluid. This fluid comes out through the microphyle in the form of a ‘pollination drop’.
Thus, some of the airborne pollen grains come in contact with the fluid and are sucked into the pollen chamber through the micropylar canal. The pollen grains are then deposited and concentrated within the pollen chamber as a result of drying off the fluid (Fig. 1.20G). At this stage, the ovule increases in size.
Fertilisation:
The sperms and the cytoplasm of pollen tube are released in the archegonial chamber by the rupture of the basal end of pollen tube. The osmotically rich pollen tube cytoplasm causes the rupture of neck cells. The motile sperms enter into the archegonium with a forward and circular motion, ciliary band forming the anterior end.
The archegonial chamber is flooded with the fertilisation fluid produced by nucellar cells. The ciliary band of the sperm is left behind on the top of the egg cell. The sperm nucleus fuses with the egg nucleus and thus a zygote in formed.
Embryogeny:
The zygote enlarges considerably and undergoes numerous free nuclear divisions. As much as 256 (C. revoluta) to 512 (C. circinalis) free bryo. The cell formation in the proembryo begins nuclei have been reported. A large central from the basal part and extends up the periphery, vacuole is formed and the nuclei are arranged.
The entire embryo never becomes cellular. The around the central vacuole (Fig. 1.22A). Most of basal cells are smaller with dense cytoplasm the free nuclei move to the base of the proem- forming the embryo (Fig. 1.22B).
A dicotyledeous embryo is developed at the tip. The upper cells elongate greatly to form the suspensor which may be coiled and twisted (Fig. 1.22C). Several egg cells may be fertilised to form many zygotes, but ultimately only one embryo in an ovule attains maturity. The layer of cells covering the outer curved face of the embryonal mass is called the cap.
Seeds:
Cycas seeds are fleshy and remain covered with an orange red-coloured thick seed coat. The embryo in the seed enlarges much. The nucellus is used up and the seed is covered with the three-layered seed coat (Fig. 1.22D). The coleorrhiza is the first region to differentiate in the embryo proper which is quite hard. The plumule develops later inside the base of the cotyledons. Subsequently the suspensor dries up.
The growth of the embryo in seeds is very slow, taking over a year to mature. Cycas shows three years reproductive cycle where pollination in first year winter, fertilisation in second year summer and seed shedding in third year summer. The development of embryo continues even after the seeds are shed.
The seeds are dispersed by birds due to their attractive fleshy outer pulp. The germination of the seed is epigeal. The first leaves come out at right angle to the cotyledons in a decussated fashion, although the remaining leaves develop spirally (Fig. 1.23).