ADVERTISEMENTS:
In this article we will discuss about the process of development of leaf, explained with the help of diagrams.
In the shoot system of plants the leaf is the principal lateral appendage of stem. A leaf has determinate growth. The foliage leaves are of two types — microphyllous- and macrophyllous leaf and the leaves are also called microphyll and megaphyll respectively. The microphyllous leaf with a few exceptions has a single vein running from base to apex. The vein is not branched. In contrast the macrophyllous leaf has veins that are branched. The venation may be open and closed.
As regards phylogenetic origin there are two theories — Telome- and Enation theory. The former states that the leaves originated from telome trusses. A telome truss consists of telome and mesome. The ultimate dichotomy of fossil plants like Rhynia, Hornea etc. is called telome. The stalk that holds the telome or internode below the telome is called mesome. Telome and mesome together constitute telome truss.
ADVERTISEMENTS:
Zimmermann in 1930 hypothesized the telome theory and it is well illustrated in the literature of Stewart (1983). They were of opinion that the primitive land plants were much like Rhynia, composed of leafless dichotomously branched stem. Later the hypothetical plant branched and the branching was in three dimensions. All branches were more or less equal in length.
During evolution some of the branches grew more rapidly than the other branches —a phenomenon called ‘overtopping’. The other slowly growing branches orient themselves in one plane — a phenomenon called ‘planation’. Later photosynthetic tissues developed between telome and mesome — a phenomenon called ‘webbing’ (Fig. 26.1).
Finally the hypothetical plant consisted of one or several main shoots, each supporting photosynthetic side branches. This type of plant is exhibited by the fossils Pseudosporochnus nodosis and Cladoxylon scoparium.
ADVERTISEMENTS:
According to telome theory macrophyllous leaf originated from telome trusses. Telome trusses oriented themselves in one plane and fused with similar lateral members. Thus a leaf with a petiole and blade is formed. The blade shows open dichotomous venation.
The lycopodian microphyllous leaf with a single vein, according to telome theory, originated from telome truss. The telome truss reduced to a single telome. One arm of dichotomy had determinate growth that formed the microphyllous leaf. The other arm of dichotomy overtopped. The leaf has a single unbranched vein (Fig. 26.2).
According to enation theory the microphyllous leaf originated as lateral simple outgrowth (enation) of stem. The fossil plants Sawdonia and Asteroxylon exhibit the enations. The enations were just minor projections. During evolution the enations increased in surface area for photosynthesis and developed stomata. At first these outgrowths had no vascular connection.
Later there appeared vascular strand that ran to the base of leaf. Still later there appeared vascular connection that ran to the tip of leaf (Figs. 26.3 & 26.3A). The microphyllous leaf is about 5 to 10mm in length and it is their maximum length. Stewart published that only Lycopodiales and the fossil group Drepanophycales have them.
 Whatever may be the interpretations of phylogenetic origin, a leaf like stem and root shows the dermal, ground and vascular tissue systems. In centric leaves the dermal system consists of epidermis. In dorsiventral and isobilateral leaves the dermal system consists of upper- and lower epidermis.
Whatever may be the interpretations of phylogenetic origin, a leaf like stem and root shows the dermal, ground and vascular tissue systems. In centric leaves the dermal system consists of epidermis. In dorsiventral and isobilateral leaves the dermal system consists of upper- and lower epidermis.
The ground tissue comprises mesophyll tissue. In dorsiventral leaf mesophy.ll is differentiated into palisade tissue and spongy parenchyma. The former occurs below upper epidermis and the later lies between the lower epidermis and palisade tissue. Vascular tissue system comprises vascular bundles that are embedded in the mesophyll tissue and form the veins.
Each of the tissue systems has separate meristem. The meristems according to position are called apical-, adaxial-, marginal-, plate- and intercalary meristem. The above meristems occur in a leaf primordium and can be observed in longitudinal and transverse sections (Figs. 26.4 & 26.5).
Leaf primordia are formed on the flanks of shoot apical meristem. They have definite sites of origin on the shoot apex according to the phyllotaxis of plant. The shoot apex fluctuates in shape and size during a plastochron- the time interval between the formation of one leaf primordium and the initiation of next leaf primordium.
Schmidt 1924 postulated that the shoot apical meristems of gymnosperm and angiosperm consist of two distinct zones —tunica and corpus. Tunica is the surface layer and it encloses the corpus. The tunica may consist of one layer (monostratose) or two to four (multistratose), the former being most common.
The corpus always consists of many layers. These two zones — tunica and corpus are distinguished on the basis of orientation of cell division. The chief mode of cell division of tunica is anticlinal irrespective of monostratose or multistratose. In anticlinal division the cell walls are laid down perpendicular to the surface of tunica.
ADVERTISEMENTS:
Anticlinal division causes the tunica to grow as a sheet but not in thickness. In contrast, corpus in addition to anticlinal division, has division with a periclinal wall, i.e. the cell wall is laid down parallel to the surface of tunica. So the cells of corpus can divide in any direction; as a result it can grow in three dimensions in contrast to tunica that can grow in two dimensions only.
The initiation of leaf commences with a lateral protrusion on the apical meristem. This is leaf buttress. Later leaf primordium develops on leaf buttress. The initial lateral protrusion results from periclinal division. The two growth zones —tunica and corpus variously participate in the development of leaf primordium.
In monostratose tunica the first divisions to form leaf occur in corpus (e.g. Scrophularia nodosa). In multistratose tunica (e.g. Vinca minor) the initial divisions occur in the innermost layer of three-layered tunica. Tunica and corpus both are involved in the initial divisions in the development of Acacia phyllode though tunica is multistratose.
Generally in most dicotyledons the first periclinal divisions occur in the subsurface layers, i.e. cells of one or more layers below. The surface layer in dicotyledons does not form the inner tissue of a leaf. It divides by its characteristic anticlinal divisions along with the growth of leaf primordium, which originates as a result of periclinal divisions of inner tissues. Protoderm of a young leaf differentiates from the surface layer.
ADVERTISEMENTS:
In gramineae and many other monocotyledons the tunica is monostratose and some members have two-layered tunica. Whatever may be the number of layers the tunica divides periclinally. As a result the main portion of leaf primordium develops from cells situated at surface and subsurface layers of the shoot apex.
Variations are also noted in the initiation of leaf primordia in gymnosperm. In Zamia and in many other conifers periclinal divisions occur in the surface and subsurface layers of the apex. In Taxodium distichum, during the initiation of leaf primordium, periclinal divisions occur in the subsurface layer of the apex. The surface layer divides with anticlinal walls.
Cunninghame et al. (1986) studied the relationship between the distribution of periclinal divisions in the shoot apex and leaf initiation in Pisnm and Silene using light and electron microscope. They concluded that periclinal divisions in the tunica and corpus, mainly in the latter cause the formation of leaf primordia.
With the aid of periclinal cytochimeras, Dermen (1947,1951) was able to detect in the apical meristem three layers of cells in Vaccininm (cranberry) and Pyrus malus (apple).
ADVERTISEMENTS:
Biseriate tunica and the peripheral layer of corpus composed the three layers. All the three layers take part in the formation of leaf. The surface layer of tunica forms the epidermis of leaf entirely by anticlinal divisions. Second layer of tunica and the peripheral layer of corpus contribute the rest of tissues of leaf.
These tissues later differentiate into photosynthetic tissues (mesophyll) and vascular tissues (vascular bundle). So the cells of tip and margin of leaf are the derivatives of surface layer of tunica. The tissues, present at the central part of leaf, i.e. between the two epidermises, are the derivatives of the inner layer of tunica and all the layers of corpus.
The periclinal divisions of superficial tunica in monocotyledons and the periclinal divisions of the subsurface layers of dicotyledons form the leaf primordium. Further divisions of adjacent and underlying cells usually follow the initial periclinal divisions. As a result the leaf primordium is elevated above the surface of the apical meristem.
Then several meristems, mentioned previously, originate in the leaf primordium and function either simultaneously or sequentially, and contribute tissues in the growth of a leaf primordium to form leaf. The growth includes apical growth, intercalary growth, marginal growth, adaxial growth and lateral growth.
Apical growth occurs by the activity of apical meristem. In vascular cryptogams an apical meristem is formed at the tip of leaf primordium and it has prolonged activity. In ferns like Lygodium the apical meristem can function for an extended period. In seed plants, though there occurs the apical meristem, the apical growth ceases relatively early.
Exceptions are noted in pinnate leaves of Gaurea (Meliaceae) where prongled apical growth occurs like Lygodium. The development of young primordium and the subsequent extension of leaf occur by cell division throughout the young primordium. Growth of leaf ceases first at the tip and last at the base. The majority of leaf growth in length is brought about by intercalary growth.
ADVERTISEMENTS:
Intercalary meristem occurs at the base of leaf primordium of most monocotyledons. The intercalary meristem present in the developing leaf primordium of Nuphar lutea causes the elongation of petiole and thus the lamina is raised above the water surface. The leaf of Narcissus elongates due to intercalary meristematic activity of leaf primordium. In this leaf apparently there is no apical growth.
Leaf primordium manifests apical growth during second plastochron; ex. Oryza sativa. At this stage the leaf primordium appears as short cone or just a small protrusion projecting from the apical meristem. Lyndon (1968) estimated the increase in length of leaf primordium of Pisum and it was about 60µm in one plastochron.
During the elongation of leaf primordium it is frequently observed that the young primordium bends over the apical meristem. This is due to greater vertical growth on the abaxial side of leaf primordium, i.e. side away from the shoot apex.
Greater growth also occurs on the two lateral sides of simple, young and dorsiventral leaf primordium. As a result small bulges or outgrowths appear on the two lateral sides. The outgrowths form wing-like strips on the margins of leaf primordium and this growth pattern is usually referred to as marginal growth.
Due to marginal growth the lamina of a leaf is formed. The marginal growth does not occur in the basal portion of leaf primordium from where the petiole is destined to develop. In Oryza marginal growth is not distinct in leaf primordium and in Narcissus there is no distinct marginal growth in the leaves.
Marginal growth occurs by the activity of marginal meristem. Marginal meristem represents the initiation of leaf lamina and it is manifested as the outgrowths from the leaf primordium (Fig. 26.5). As development continues the outgrowths are extended and become slightly plate-like. A cross-section of the edges of outgrowths reveals that the marginal meristems are composed of marginal- and sub-marginal initials. Foster (1936) illustrated the future destiny of the derivatives of the initials (Fig. 26.6).
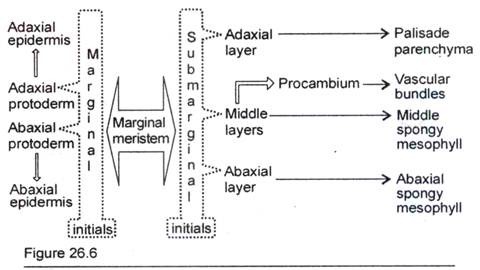 Diagram illustrating the cell lineages involved in the development of leaf of Nicotiana tabacum.
Diagram illustrating the cell lineages involved in the development of leaf of Nicotiana tabacum.
In young lamina the outermost cell layer of margin is the marginal initial. The marginal initials form the protoderm. Abaxial and adaxial protoderm layers both are the derivatives of marginal initials. Generally in angiosperms the marginal initials divide anticlinally, i.e. cell walls are laid down perpendicular to surface of initials.
So the protoderm layers grow as a sheet, but not in thickness. Foster (1937) and Sharman (1942) reported the occurrence of periclinal divisions in marginal initials of bud scales of Rhododendron and in certain monocotyledons. In Prunus, Pelargonium, Dracaena and Veronica the leaves are variegated.
In these leaves the marginal initials form protoderm. Protoderm divides periclinally and forms the white margins of leaves. The marginal initials produce the whole lamina in Eubrachion of the family Viscaceae and Daphne of the family Thymelaeaceae.
The cells situated just below the marginal initials are the sub-marginal initials. These initials divide in various planes and donate cells to the inner layers of developing lamina. The sub-marginal initials, after divisions, establish the number of layers of cells in the lamina. The future destiny of the derivatives of sub- marginal initials is much variable and a few are illustrated in Fig. 26.6-26.9.
In Xanthium the marginal growth is initiated when the leaf primordium is at about 0.22 mm long. The activity of marginal meristem is continued for about 23 days. As a result six layers of cells are established in the leaf. The young leaf primordium when observed in its cross-sectional view reveals the number of cell layers and the occurrence of marginal meristem at the edges of the flanges. The flanges consist of the tissues that project from the future midrib.
The leaf marginal meristems with marginal and sub-marginal initials resemble shoot meristems with tunica and corpus. The marginal initials are equivalent to tunica whereas the sub-marginal initials represent the corpus. This analogy is based on the patterns of cell division. The marginal initials and tunica generally divide anticlinally. The sub-marginal initials and corpus divide in various planes.
The leaf primordium of many dicotyledons has adaxial growth. This growth is due to the activity of adaxial meristem, Fig. 26.4C. Adaxial meristem originates as a strip of cells below the adaxial epidermis. The chief mode of division of adaxial meristem is periclinal. The cells, thus formed, contribute to the thickness of leaf especially to growth in thickness of the midrib and petiole.
The phyllode of Acacia and the leaf of Acorns are flattened laterally instead of dorsiventrally. The adaxial meristem in these plants is extremely active. As a result the laterally flattened lamina-like structure is formed. Moreover in both genera marginal growth is suppressed. It favours the activity of adaxial meristem and so the foliar organ extends in median plane.
In the early stages of development the activity of marginal meristems establish a certain number of layers of cells in the leaf lamina. These cells divide anticlinally, i.e. at right angles to the surface thus forming ‘plate’ of cells. These cells are referred to as plate meristem (Fig. 26.4D), the activity of which causes the expansion of lamina laterally.
The whole lamina functions as a plate meristem and the derivative cells contribute to lateral growth. The leaf marginal meristems do not function indefinitely. So the leaves have a limited and determinate width. The marginal meristem forms definite number of layers of cells. These derivative cells are the plate meristems and consist basically the entire lamina.
As the cells of the lamina divide anticlinally the layer of cells grow in surface area but not in thickness. Though the cell division in plate meristem is predominantly anticlinal, exception is noted in Xanthium where some periclinal divisions do occur thus forming additional cell layers and causing growth in thickness of the leaf.
In the developing lamina the marginal meristem forms a certain number of layers of mesophyll cells. Usually the adaxial layers of mesophyll give rise to palisade tissues.
The abaxial layers of mesophyll form the spongy cells. The middle layer of cells situated between adaxial and abaxial layers form the procambium of veins. In some cases spongy cells may develop from parts of middle layer. It is to note that the layer, which is destined to form procambium, varies according to plants.







