ADVERTISEMENTS:
The general concepts of the biotechnological approaches for the abatement of pollution with special reference to the following aspects are described hereunder :
i. Atmospheric CO2 reduction.
ii. Sewage treatment by bacteria and algae.
ADVERTISEMENTS:
iii. Eutrophication and phosphorus pollution.
iv. Management of metal pollution.
v. Immobilized cells in the management of pollution.
i. Atmospheric CO2 Reduction:
The lower atmosphere contains CO2 at a concentration of 0.0314% by volume. There is a continuous addition of CO2, particularly coming from industrial processes. Any increase in the content CO2 has to be viewed very seriously, since it is the principal gas that causes greenhouse effect and rises atmospheric temperature.
ADVERTISEMENTS:
It is suggested that the global temperature will increase by around 2-5°C on doubling the atmospheric concentration of CO2. The present belief is that during the past 100-150 years, the CO2 level increased by about 25% with an increase in the atmospheric temperature by about 0.5%. Thus, CO2 is closely linked with global warming.
For the reasons stated above, the reduction in atmospheric CO2 concentration assumes significance.
There are mainly two approaches for the biotechnological reduction of CO2 in the atmosphere:
1. Photosynthesis
2. Biological calcification.
Photosynthesis to Reduce Atmospheric CO2:
Utilization of CO2 for photosynthesis by plants is the most significant process to reduce CO2 content in the atmosphere. Photosynthesis may be represented by the following equation.
ADVERTISEMENTS:
Higher plant photosynthesis:
The fast growing trees are more efficient in utilizing CO2 for photosynthesis, hence their propagation is advocated. Further, micro- propagation and synthetic seed production through plant tissue culture techniques are also important. The major problem is the continuous deforestation that is associated with a possible increase in atmospheric CO2. The conservation of forests and plantations is the need of the hour.
Certain biotechnological approaches have also been made to improve the CO2 utilization by enhancing photosynthesis. The enzyme ribulose- bisphosphate carboxylase (RUBP-case) is closely linked with CO2 fixation. There are some attempts to genetically manipulate this enzyme so that the photosynthetic efficiency is increased.
Microalgal photosynthesis:
ADVERTISEMENTS:
Certain microalgae are more efficient than higher plants in utilizing atmospheric CO2 for photosynthesis e.g. Chlorella pyrenodiosa, Spirulina maxima. These organisms are capable of generating more O2 than the amount of CO2 consumed.
It is advocated that growing microalgae in the vicinity of industries and power plants where the production CO2 is very high will help to minimise the polluting effects of CO2. In addition, these microalgae, appropriately regarded as photo bioreactors, accumulate as biomass which can be utilized for the extraction of protein for use as food or feed.
Genetic manipulations are in progress to develop new strains of microalgae that can tolerate high concentrations of CO2, and yield larger biomass. Some success has been reported in the mutants of Anacystis nidulans and Oocystis sp.
Biological Calcification to Reduce Atmospheric CO2:
ADVERTISEMENTS:
Certain organisms present in the deep sea (corals, green and red algae) are capable of storing CO2 through a process of biological calcification. The overall process of calcification may be represented as follows.
H2O + CO2 → H2CO3
H2CO3 + Ca2+ → CaCO3 + CO2 + H2O
As the CaCO3 gets precipitated, more and more atmospheric CO2 can be utilized for its formation.
ii. Sewage Treatment by Bacteria and Algae:
ADVERTISEMENTS:
The waste water resulting from various human activities (domestic), agricultural and industrial is technically referred to as sewage. The sewage is mostly composed of organic and inorganic compounds, toxic substances, heavy metals and pathogenic organisms.
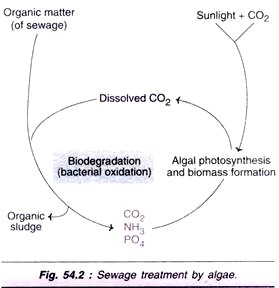
In the biological treatment of sewage, the organic matter is subjected to biodegradation or bacterial oxidation. In this way, the organic matter is degraded to smaller molecules (CO2, NH3, PO4 etc.), Biodegradation requires a constant supply of O2. This can be done by continuous bubbling of atmospheric O2 through a specially designed equipment. This is an expensive process, besides the involvement of manpower.
A continuous supply of O2 can be achieved by growing microalgae in the ponds where sewage treatment is being carried out. The algae are very efficient in photosynthesis and they release O2 in to the ponds. This is the form of dissolved O2 (Fig. 54.2). Thus, algal-bacterial symbiosis is responsible for photosynthetic oxygenation and the biodegradation of sewage organic matter. This is a natural and inexpensive process.
In addition to the supply of CO2, the algae used in sewage treatment can adsorb certain toxic heavy metals also. This is possible due to the negative charges on the algal cell surface which can take up the positively charged metals. The algal treatment of sewage is useful in several other ways.
i. Support fish growth, as the algal cells are good food for fishes, besides supplying dissolved O2.
ADVERTISEMENTS:
ii. The algal cells, rich in protein, serve as good food and feed.
iii. Certain metals (that are adsorbed from the sewage) can be recovered.
iv. The load of pathogenic microorganisms is very much reduced (they either die or settle at the bottom of the pond).
The algal species of different genera ideally suited for sewage treatment are Chlorella, Euglena, Chlamydomonas, Scenedesmus, Ulothrix and Thribonima.
iii. Eutrophication and Phosphorus Pollution:
Sewage and waste waters from industries and agriculture are rich in organic and inorganic nutrients, containing phosphorus and to a lesser extent nitrogen. These nutrients favour excessive growth of algae which results in oxygen depletion (de-oxygenation) and this phenomenon is referred to as eutrophication. Eutrophication results in the death of non-resistant organisms (e.g. fishes), and foul smell.
ADVERTISEMENTS:
Water blooms:
Certain species of algae (particularly blue green algae e.g. Lynhbia, Microcystis, Oscillatoria, Anabaena) that can tolerate eutrophication, grow well and form water blooms in waste/sewage water. From the extensive formation of water blooms, one can predict that pond has accumulation of very high quantities of phosphorus.
The algae are capable of absorbing phosphorus, and store them as polyphosphates. Phosphorus is a strong promoter of algal growth and water bloom formation. Water blooms are associated with the production of certain toxins (e.g. lipopolysaccharides) that are harmful to fishes and birds. These toxins may also cause certain diseases in humans e.g. diarrhea, gastroenteritis, nausea.
Control of eutrophication:
There are broadly two ways of controlling algal growth resulting in eutrophication. In the chemical approach, algicides such are copper sulfate, sodium arsenate and 2, 3-dichloronaphthoquinone are used. But the chemical treatment is associated with increase in sludge volume. In the biological approach, cyanophages (i.e. the viruses that can kill algal cells) can be used to contain eutrophication.
Biological Removal of Phosphorus:
As stated above, phosphorus significantly contributes to algal growth in waste water. Removal of phosphorus therefore can protect from eutrophication. It is possible to precipitate phosphorus with salts of calcium, magnesium and aluminium. This however, is not practicable on large scale. In addition, chemical methods add to the sludge volume. Hence, biological methods are preferred.
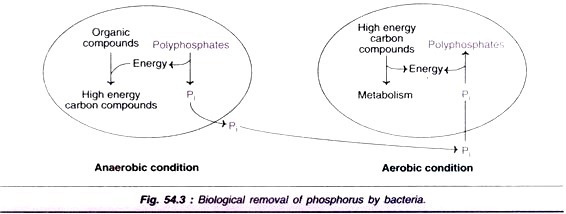
Biological removal of phosphorus by bacteria is carried out through aerobic and anaerobic processes, as depicted in Fig. 54.3. Under anaerobic conditions, certain organic compounds (e.g. acetate) obtain their energy from polyphosphates to store as carbon reserves. In this process, free inorganic phosphorus is released.
In the subsequent aerobic process, the organic carbon reserve compounds are oxidized to generate energy. A small portion of this energy is utilized for the conversion of phosphates to polyphosphates. The net effect is that phosphorus accumulates in the bacterial cell in the form of polyphosphates.
iv. Management of Metal Pollution:
Environmental pollution with heavy metals (e.g., lead, cadmium, mercury) causes several toxic manifestations in living organisms, including cancer. Bioaccumulation, bio-magnification and bio-methylation are some of the characteristic features of metal pollution.
As the living organisms (including man) are constantly exposed to metals, they accumulate by a process referred to as bioaccumulation. Continuous exposure and accumulation of a given metal in the organisms results in its increased concentration, a phenomenon referred to as bio-magnification. Bio-magnification usually occurs through food chain and man is the ultimate victim.
Bio-methylation is the process of transfer of methyl groups from organic compounds to metals. This is carried out by microorganisms in the soil and water. Although it is true that some metals get detoxified by methylation (e.g. arsenic), some of them may even become more toxic (mercury).
Bio-scavengers of Metals:
Certain aquatic plants namely phytoplankton’s (plants that freely float on water surface) or benthics (plants attached to some substratum at the bottom of aquatic reservoirs) and microorganisms can take up the metals from the bathing media (waste water, ponds) and act as natural bio-scavengers. Besides combating the pollution effects of metals, this process is also useful for the recovery of industrially important metals from the bio-scavengers.
In the recent years, environmental protection agencies (EPAs) world over are advocating the use of aquatic plants and microorganisms as low cost bio-absorbents for the removal of toxic metals from the environmental pollution. Some of the plants and microorganisms useful for the management of metal pollution are briefly described.
Plants in metal pollution management:
Water hyacinth:
This is the most common weed found in tropical waters and its control is a major problem. This plant can absorb and accumulate cadmium, mercury, lead and even silver to a significant extent.
Water lettuce:
This is a floating aquatic weed, and is also a good metal bio-scavenger. It can take up large quantities of lead, cadmium, mercury and arsenic.
Cat-tails:
These plants possess long erect leaves and grow in marshy areas. Cat-tail leaves have been shown to be useful for the absorption of nickel, copper, besides calcium and magnesium salts. Water spinach that accumulates lead and chromium is eaten in some areas by poor people. The toxic metals are certainly toxic to humans.
Ferns:
The fern namely Azolla filicoides accumulates cadmium, copper and uranium. Salvinia sp can absorb several metals including lead, nickel and chromium.
Microorganisms in metal pollution management:
Algae:
Several metals from the fresh water can be absorbed by algal blooms. For instance, Chlorella vulgaris can take up copper, mercury and uranium. The marine microalgae (e.g., Ulva, Laminaria) are also useful for the abatement of metal pollutants.
Fungi:
Certain fungal species are good absorbers of heavy metals (Pb, Hg). e.g. Rhizopus, Aspergillus, Penicillin, Neurospora.
Bacteria:
A few bacterial species can accumulate metals on their cell walls. For instance, E. coli can take up mercury while Bacillus circulans can accumulate copper. Attempts are being made to improve the strains of bacteria (through genetic manipulations) for more efficient uptake and accumulation of toxic metals. Further, such species will also be useful for the recovery of metals from the bacterial biomass.
Mechanisms of Metal Scavenging:
The aquatic plants and microorganisms may take up the metal pollutants through absorption and/or adsorption. The mechanism of accumulation of metals by the bio-scavengers is very complex and may involve one or more of the following processes.
Binding of metals to cell walls:
Bioaccumulation of metals on the cell walls is commonly observed in lower plants-algae, fungi and bacteria. Even some higher plants can concentrate metals on the cell walls. There is some evidence to’ show that metals are held to certain organic biopolymers on the cell walls, till the binding sites get saturated.
Accumulation of metals in cellular compartments:
Plants are capable of transporting metals to intracellular and intercellular free spaces, and the cellular organelles (lysosomes, vacuoles). Certain metals occur as immobilized metal containing crystals. For instance, heavy metal complexes of calcium oxalate crystals have been detected in algae.
Synthesis of metal binding proteins:
Several algae and certain fungal species are known to synthesize metal binding proteins or peptides. Phytochelatin is an ubiquitous protein present in all the plants. It is a metal chelating protein and is regarded as a common buffering molecule for the homeostasis of metals.
Phytochelatin is rich in cysteine and can form salt metal complexes through sulfhydryl (SH) groups. It is comparable in its functions to metallothionein found in animals. Certain metals are known to induce the synthesis of phytochelatin. Some workers, in recent years, have suggested phytochelatin can be used as a biomarker for metal pollution detection.
v. Immobilized Cells in the Management of Pollution:
The use of immobilized cells, particularly microbial whole cells, for the abatement of environmental pollution is a recent development. The general aspects of cell immobilization, techniques involved, and their applications are described elsewhere the immobilized cells are useful for the waste water treatment, and for the recovery of metals from industrial effluents.
A selected list of the pollutants and the immobilized microorganisms used for their abatement is given in Table 54.2. For instance, phenol and triethyl lead can be removed respectively by immobilized Pseudomonas putida and Arthrobacter sp.
For more efficient management of a group of environmental pollutants simultaneously, immobilized systems with more than one type of microorganisms are used. In the recent past, genetically engineered microorganisms (OEMs) are being developed for the treatment of polluted water and soils.