ADVERTISEMENTS:
Water pollution is a major environmental problem.
The main desirable uses of water are:
(a) Municipal water supply,
(b) Industrial water supply,
ADVERTISEMENTS:
(c) Recreational uses such as swimming, boating, aesthetics,
(d) Agriculture and fisheries, and
(e) Ecological balance.
For each of these uses there are certain water quality requirements. Major water quality problems arise through interference with various uses of the water. These problems are confirmed through water quality monitoring and analysis. For example, the problem of low dissolved oxygen (DO) content in a stream interferes with the fish life of that stream besides promoting growth of pollution tolerant organisms and disappearance of clean water species, and the manifestation of that interference is with foul odours. Water Pollution sources are generally described as point sources and non-point sources.
ADVERTISEMENTS:
A point source is any single identifiable source of pollution from which pollutants are discharged, e.g., the outlet pipe of a municipal wastewater treatment plant or industrial discharge. A nonpoint source is more diffuse and includes a variety of wastes from streets, construction sites, agriculture and food processing and mining wastes.
Water Pollutants:
Water is polluted by:
(i) Sewage and domestic wastes,
(ii) Pesticides,
(iii) Detergents and synthetic fertilizers,
(iv) Toxic metals and pollutants,
(v) Oil, grease and other chemicals,
(vi) Acid deposition (increased pH),
ADVERTISEMENTS:
(vii) Suspended solids,
(viii) Thermal pollution, and
(ix) Pathogenic microorganisms.
Wastewater Treatment:
ADVERTISEMENTS:
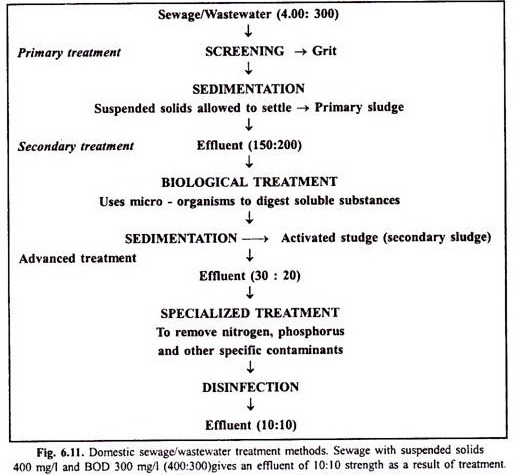
Wastewater (or sewage) treatment is done in three stages – primary, secondary and advanced. Primary treatment first removes large objects through screening, and then smaller objects such as sand and small stones. Suspended solids remain in the wastewater, which is settled out in a sedimentation tank. After removal of solid materials, the wastewater is given secondary treatment (Fig. 6.11), where micro- organisms digest soluble contaminants.
However, phosphorus and nitrogen are not removed by this process and we require advanced or tertiary treatment for this purpose. The major function of tertiary treatment is to reduce the load of phosphorus and nitrogen through precipitation, nitrogen stripping, carbon absorption and chlorination. Two most common additional methods of tertiary treatment are physicochemical and advanced biological treatment.
The physicochemical treatment includes the addition of reagents to wastewater to precipitate various entities, as well as carbon adsorption columns, ion exchange and reverse osmosis. It is especially suited to industrial application such as those involving heavy metals, and to wastes with very low or very high pH.
ADVERTISEMENTS:
In advanced biological treatment, wastewater is treated by biological communities, such as water hyacinths, after secondary treatment and before it is discharged into receiving waterways. However, due to cost factor advanced treatment is not always used. Usually the cost of treatment of wastewater doubles from primary to secondary.
The advanced treatment plants are twice as expensive to build as secondary treatment plants and probably four times as expensive to operate. Obviously, the cost of membrane separation (reverse osmosis), though quite useful for industrial wastewater treatment, is exorbitantly high. In areas where sewer lines are not available, the traditional septic tanks are used for sewage and wastewater treatment. These systems produce a concentrated septage, which must be periodically pumped out and, if possible, taken to a municipal plant for treatment.