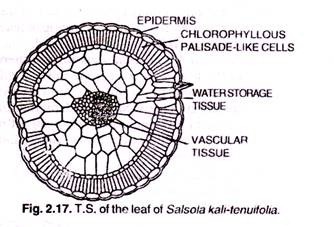
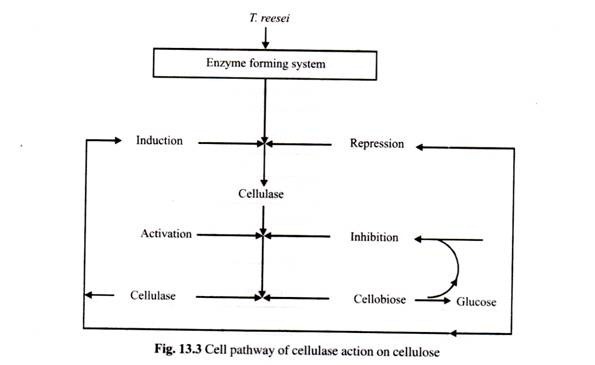
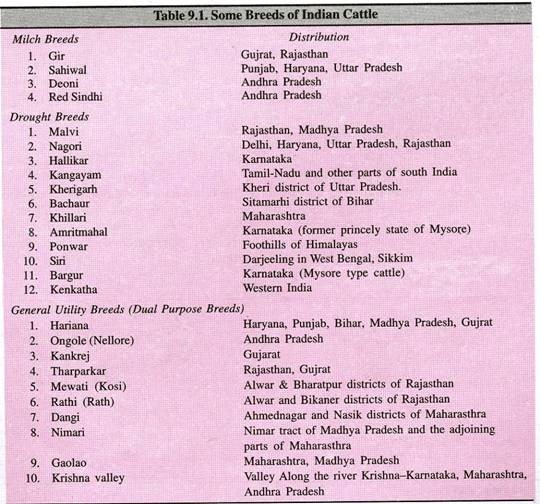
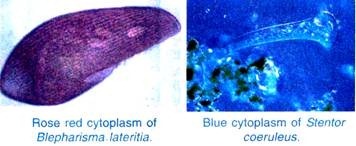
ADVERTISEMENTS:
Cancer is a disease characterized by the excessive and abnormal growth of certain tissue cells.
In the healthy tissues of an adult, growth is regulated such that cell proliferation exactly balances the rate of cell loss. As a result, once one reaches adult age, the sizes and cellular contents of the various body organs remain more or less constant.
Various chemical, physical, and viral agents can cause the loss of normal control of cell growth; as a result, a normal cell may be transformed into a cancerous one.
ADVERTISEMENTS:
The unregulated cell growth that accompanies cellular transformation produces tumors or neoplasms, each tumor being the product of proliferation of a single aberrant cell.
Malignant tumor cells are cancer cells that spread to and take up residence in neighboring tissues—a condition called metastasis. On the other hand, benign neoplasms consist of cancer cells that do not spread to distant sites and do not pose so great a threat to life. The body’s major line of defense against cancer cells is the immune system, which recognizes the changes in cell surface antigens that frequently accompany cellular transformation. As a result, nearly all cancer cells are eliminated by the immune system before causing serious harm.
Cellular transformation can occur in vitro (i.e., in cultures of cells). Cultures of normal mammalian cells such as fibroblasts have a finite life span. Usually such cultures can be maintained for no more than 40 or 50 generations, after which the cells become senescent and the culture dies out. In vitro transformation may convert a normal culture into an “immortal” one—one that can be indefinitely sub- cultured. The relative ease with which cancer cells can be grown and maintained in culture has been a boon to the study of cellular transformation.
Transformed fibroblasts, like cancer cells in vivo, exhibit abnormal (i.e., excessive) growth. Normal fibroblasts in culture are flattened cells that exhibit contact-inhibited cell growth once they form a single layer. Cancerous fibroblasts grow more rapidly, form clumps of cells, do not exhibit contact inhibition, and will grow on top of one another. In normal cultures, the cells are anchorage-dependent; cancerous cells are anchorage-independent.
ADVERTISEMENTS:
When embryonic kidney or liver cells are grown in culture, they adhere to one another. It is as though they recognize their neighbors as cells of the same type and form the intimate associations’ characteristic of kidney and liver tissue. When kidney and liver cells are intermixed, they sort themselves out into regions that consist of one or the other cell type.
Transformed cells exhibit diminished adhesion properties (apparently as the result of a change in the glycoprotein and glycolipid content of the plasma membrane). Such changes explain why cancer cells dissociate from the tissues from which they originate and are able to spread (i.e., metastasize) to other areas of the body and grow.
Mutagenesis, Proto-oncogenes and Oncogenes:
The growth of a cancer stems from changes that occur in a single (and previously normal) cell. The transformed cell escapes immune detection and undergoes uncontrolled growth and division producing a large clone of transformed cells that is now identified as a tumor. Among the agents that have been shown to be carcinogenic (i.e., “cancer-causing”) are various chemicals, radiation, and infection by certain viruses. Carcinogens transform normal cells by directly or indirectly causing changes in the cell’s DNA. These changes are called mutations, and the carcinogen is thus said to be mutagenic.
It is now clear that most (perhaps all) cancers stem from the translocation, alteration, or activation of specific genes called proto-oncogenes. Proto- oncogenes are believed to be present in all normal cells, where they exist either in an inactive state or function in the production of normal quantities of transcription and translation products. Mutations change or activate proto-oncogenes and convert them to oncogenes, whose translation products (i.e., oncoproteins) are intimately associated with the cancerous state.
Cancer of the Immune System—Burkitt’s Lymphoma:
Some cancers are clearly associated with changes in the numbers of chromosomes in the nucleus. Normal human somatic cells contain 46 chromosomes (i.e., 23 homologous pairs), whereas cancer cells have irregular numbers—a condition termed aneuploidy. For example, HeLa cells (a cancer cell line maintained in subculture for many years and originally derived from a woman with cervical cancer) contain 70-80 chromosomes.
Indeed, it is typical for there to be more than the normal chromosome number in cancer cells, but it is generally agreed that the aberrant number is the result of uncontrolled cell growth and not the cause of it. More subtle chromosome changes can be linked to cancer causation. One of the best illustrations of this is the human leukemia called Burkitt’s lymphoma.
We saw that the remarkable diversity of antibodies produced by B cells results from the translocation of gene segments during cellular proliferation. In the most common forms of Burkitt’s lymphoma, a reciprocal translocation occurs between chromosomes 8 and 14 (Fig. 25-19).
A segment at the end of chromosome 8 is trans-located to chromosome 14 and is replaced by a segment from the end of chromosome 14. As a result, a proto-oncogene from chromosome 8 ends up near genes of chromosome 14 that encode part of an antibody molecule. This translocation serves to convert the proto-oncogene to an oncogene. The oncogene is called myc because the gene itself was first identified in a virus known as MC29 (myelocytomatosis virus).
The myc gene consists of three exons and two introns (Fig. 25-19). The introns consist of DNA that is not transcribed into mRNA and is not expressed as protein. However, the first exon (which is about 650 base pairs long) also does not code for protein; rather, protein synthesis begins with the second exon.
In the most common forms of Burkitt’s lymphoma, either all three exons are translocated from chromosomes 8 to chromosome 14 or exons 2 and 3 are trans-located (leaving exon-1 on chromosome 8). Translocation places the myc gene in a region of chromosome 14 that is adjacent to the genes encoding the constant regions of immunoglobulin heavy chains. In rarer forms of the lymphoma, the myc gene remains on chromosome 8 but it is joined there by genes translocated from either chromosome 22 or chromosome 2. Translocation involving chromosome 2 brings k light chain constant domain genes next to the myc gene; translocation involving chromosome 22 brings λ light chain constant domain genes next to the myc gene.
Apparently, translocations that result in the myc gene being next to antibody encoding genes causes a change in the myc gene’s expression. The nature of the change is not known with certainty, but it could be excessive production of the myc gene product or production at the wrong time in the cell cycle. The nature of the myc gene’s protein product is still uncertain. However, the viral myc gene product is a protein that binds to DNA.
ADVERTISEMENTS:
Naming oncogenes:
Oncogenes are usually identified using a three-letter name that is related in some fashion either to the gene’s source or to its cause. To distinguish between viral oncogenes and their native cell counterparts, the viral gene name is preceded by a “v” (for virus), whereas the cellular oncogene name is preceded by a “c” (for cell).
Cancer of the Immune System—T-Cell Leukemia:
It is now clear that a number of cancers result from infection by retroviruses.
ADVERTISEMENTS:
Two general mechanisms have been proposed to account for the capability of retroviruses to transform cells:
(1) The transfer of oncogenes from the retrovirus to the infected host cell and
(2) The activation during infection of oncogenes already present in the host.
Not all retroviruses contain oncogenes in their genome, but those that do are believed to have acquired them from the cellular DNA of a host cell during a previous infection. That is, the oncogene was a normal constituent of the host cell’s genome but was picked up by the virus during its assembly within the host. Transfer of the oncogene from the virus to a new cell during a subsequent infection leads to the transformation of that cell. Retroviruses contain repeating nucleotide sequences at the ends of their genomes; these sequences are known as long terminal repeats (LTRs).
ADVERTISEMENTS:
LTRs contain regulatory segments known as promoters and enhancers; these serve to activate transcription of viral genes into messenger RNA. Retroviral transformation of host cells by activation of oncogenes that are already present in the host cell may involve insertion of an LTR near one of the cellular oncogenes.
Two viruses, called ETLV-I and HTLV-II, are the cause of T-cell leukemia in humans. These leukemias are characterized by the excessive and abnormal production of T cells. The HTLV genome does not contain any oncogenes and appears not to transform cells by either of the mechanisms described above. One model proposed to explain the actions of the HTLVs postulates that the virus directs the infected host cell to produce a protein that stimulates not only production of new viruses but also production of new host cells.
The viral promoter has been shown to be present in the LTR and when integrated into the host genome increases the transcription of genes that turn on cell division. This then leads to the abnormal and uncontrolled growth of the transformed cells.
A close relative of HTLV-I and HTLV-II, called HTLV-III, causes acquired immune deficiency syndrome (AIDS) (see above). In contrast to the effects of HTLV-I and HTLV-II, HTLV-III causes T-cell death rather than excessive proliferation.
Viruses and Cancer:
That a link exists between tumors and viruses has been known since the beginning of the century. As early as 1910, P. Rous was able to show that an avian leukemia could be transmitted from one infected fowl to another using viral particle-containing extracts of the cancerous cells. The virus causing this cancer is known as the Rous sarcoma virus (RSV). In the 1960s, R. Dulbecco and M. Vogt were able to demonstrate virus induced cellular transformation in vitro. Dulbecco and Vogt worked with hamster embryo fibroblasts, which they cultured in Petri dishes containing agar growth medium.
ADVERTISEMENTS:
In normal cultures, cell proliferation produces a monolayer that eventually covers the surface of the dish. However, addition either polyoma virus or Simian virus 40 (SV-40) to the culture transforms the cells. Growth becomes irregular and clumps of cells raised well above the surface of the agar quickly appear. When transferred to young animals, the transformed cells gave rise to tumors.
The polyoma and SV-40 viruses are DNA tumor viruses, that is, the viral genome consists of DNA. Included in the DNA is an oncogene that becomes integrated into the DNA of the host cell and that when expressed causes cellular transformation.
More extensively studied and better understood are the RNA tumor viruses. All of the RNA tumor viruses are retroviruses. When the RNA of a retrovirus is inserted into the host cell, it is transcribed into DNA (the process is usually referred to as reverse transcription). The DNA produced by reverse transcription of tumor virus RNA includes oncogenes. Integration of an oncogene into the host cell’s genome and its eventual expression causes cellular transformation.
Oncogenes and Their Protein Products:
The first viral oncogene was specifically identified in the 1970s in the Rous sarcoma virus and was named v- src (for sarcoma). As noted above, this virus causes leukemia in chickens. The v-src oncogene encodes a protein called p60src having a molecular weight of 60,000.
In 1975 it was found that the src gene is nearly an exact copy of a gene that is found in all chickens. Apparently, the chicken proto-oncogene was picked up by a retrovirus during infection, became a part of the viral genome, and changed into an oncogene that causes cancer when reintegrated into the genome of a newly infected host cell.
ADVERTISEMENTS:
About 20 oncogenes have now been identified in retroviruses. When transferred to host cells by infection, they cause a number of cancers (e.g., carcinomas, sarcomas, and lymphomas) in birds, mice, rats, pats, and primates. In each instance, the oncogene has proven to be closely related to a normal gene of the host and to encode a protein that is similar to a normal host cell protein.
Not all tumors result from viral infection, but like virus-induced cancers, the cells of tumors of non-viral origin also contain oncogenes. The oncogenes of such tumor cells are called tumor oncogenes. DNA that contains tumor oncogenes may be isolated and introduced into normal cells. When this is done, the normal cells are transformed into cancerous ones. The phenomenon is called transfection and is a valuable tool for studying cellular transformation.
As you may have inferred by now, tumor oncogenes and viral oncogenes appear to be derived from a common gene pool. For example, the retroviral oncogene v-ras carried by the murine sarcoma virus is homologous to a tumor oncogene. The c-ras tumor oncogene is derived from the proto-oncogene proto-ras, which is present in animal species ranging from yeasts to humans. This high degree of conservation suggests that in addition to being a source of oncogenes, the proto- oncogenes play important roles in the untransformed cell.
Conversion of a proto-oncogene to an oncogene may occur in several ways. One mechanism believed to be important in the activation of oncogenes is mutation induced by a carcinogen. In 1982, R. A. Weinberg and M. Barbacid (working separately) showed that the nucleotide sequence in the T24 human bladder carcinoma oncogene c-ras differed at only one position from the nucleotide sequence of the normal, proto- oncogene.
A single point mutation in which guanosine occurs in place of thymidine results in the incorporation of valine instead of glycine at the twelfth amino acid position of the oncogene-encoded protein. This solitary substitution appears to be all that is necessary to confer cell-transforming properties to the gene product. Another indication of the cancerous state of a cell is indicated by alteration of protein kinases in the membranes of certain cells.
Protein Kinases:
Protein kinases are enzymes that phosphorylate proteins by adding phosphate to hydroxyl-containing R-groups of certain amino acids (Fig. 25-20). In normal cells, about 90% of the phosphate coupled to protein is coupled to serine and 10% is coupled to threonine.
Phosphorylation of the hydroxyl group of tyrosine is very rare (about 0.02%). In 1978, it was found that the protein encoded by the v- src oncogene (i.e., p60src) is a plasma membrane- bound protein kinase that phosphorylates tyrosine, and shortly thereafter the src proto-oncogene was also shown to encode a tyrosine-phosphorylating protein kinase.
These findings indicated that the products of viral oncogenes mimic the functions of their normal cellular counterparts. However, in transformed cells there is a 10-fold increase in the phospho- tyrosine level. It appears, therefore, that an abnormally high phosphorylation of tyrosine may be related to cell transformation.
Among the cellular proteins that are acted upon by protein kinases is vinculin, a cytoskeletal protein. In cells transformed by the Rous sarcoma virus, there is a 20-fold increase in the amount of phosphorylated tyrosine present in vinculin. Because vinculin is believed to play a role in determining cell shape, there may be a connection between the unusual shape of transformed cells and the excessive phosphorylation of cellular vinculin.
Certain metobolic pathways can also be altered in cells by changes in protein kinase levels. For instance, it has been known for many years that tumor cells produce large amounts of lactic acid as a result of increased glucose catabolism by glycolysis. Though a small fraction of three of the enzymes of glycolysis contain phosphotyrosine (e.g., enolase, phosphoglycerate mutase, and lactate dehydrogenase), the percentage is so small as to raise considerable doubt regarding a link to transformation. Moreover, the rate-limiting enzyme for glycolysis (phosphofructokinase) does not appear to contain phosphorylated tyrosine.
Growth Factors and Growth Factor Receptors:
Among other things, the growth of cells in culture can be regulated by addition of a number of secretory polypeptides called growth factors. It is believed that in vivo these factors are secreted into the blood by the cells of one tissue and interact with specific plasma membrane receptors, called growth factor receptors, in other tissues. Binding of growth factors to growth factor receptors triggers the production of intracellular signals that ultimately cause DNA synthesis and mitosis. Several oncogenes have been identified whose protein products are similar to growth factors and to growth factor receptors.
Among the known growth factors is platelet- derived growth factor (PDGF), a protein having a molecular weight of about 30,000. It has recently been found that the PDGF is nearly identical to a portion of the protein product of a viral oncogene—the v-sis oncogene of Simian sarcoma virus (SSV).
It is believed that in SSV-transformed fibroblast cells, the v-sis oncogene is expressed as a secretory oncoprotein that interacts with PDGF receptors in the fibroblast plasma membrane. Binding of the oncogene product to the PDGF receptors acts to stimulate tyrosine phosphorylation of intracellular proteins and causes unrestricted growth of the cells. In situ, normal cells bearing the PDGF receptor respond to PDGF from other cell sources. Transformed cells produce and release their own PDGF, which then acts to promote cell proliferation by binding to the cells’ PDGF receptors.
Another growth factor called epidermal growth factor (EGF) stimulates cell proliferation following binding to cellular EGF receptors. EGF receptors are large glycoproteins (MW 175,000) having a tyrosine kinase activity that is stimulated severalfold on binding EGF. EGF receptors have amino acid sequences that are closely related to the product of the viral oncogene v-erb-B.
This oncogene is present in the genome of avian erythroblastosis virus (AEV), a virus that attacks chickens and other birds. Apparently, the viral gene was derived from the normal EGF receptor gene that is present in avian cells.
Results of studies on PDGF and EGF, their receptors, and the corresponding oncogene products again implicate protein kinases with cellular transformation. One of the models proposed to account for the effects of the growth factor-receptor system is shown in Figure 25-21. The growth factor receptors are intrinsic membrane proteins that span the lipid bilayer.
The growth factor receptor site is located in that portion of the protein that faces the outside of the cell, whereas the site for tyrosine phosphorylation faces the interior. Binding of the growth factor to the receptor site serves to activate the tyrosine-specific protein kinase site, and as a result intracellular target proteins are phosphorylated. It is believed that the tyrosine-phosphorylated proteins then act in some way to promote cell proliferation.
Nuclear Oncoproteins:
The products of some oncogenes are proteins that have an effect directly within the cell nucleus. Included in this group is the myc gene discussed earlier in connection with Burkitt’s lymphoma. The c-myc oncoprotein binds to nuclear DNA and in some manner makes replication of the DNA a continuous process. Other oncogenes whose products act within the nucleus are the c-myb and c-fos oncogenes. The c-myb gene is associated with a leukemia in birds and c-fos produces a sarcoma in rats. At this time, little is known about their oncogenic protein products or their modes of action within the nucleus.
Table 25-2 lists the genes, their retroviral sources, and their supposed action. Because more than 20 oncogenes have been discovered so far, Figure 25-22 and Table 25-2 are by no means complete.
Multiple Oncogene Activation and Cancer:
The link between cancer and either the activation of cellular proto-oncogenes or the acquisition of viral oncogenes is irrefutable. Activation of cellular oncogenes may stem from exposure either to chemical mutagens or to physical mutagens (e.g., radiation).
Uncertain at the present time is whether activation or acquisition of a single oncogene is sufficient to fully transform normal cells into cancerous ones. R. A. Weinberg has shown that cells cultured immediately after removal from an animal are not easily transformed by a single oncogene, whereas culture cells from established cell lines are.
Pairs of oncogenes, such as myc and ras, appear to cooperate in some manner such that they readily transform cells cultured immediately after removal from an animal. It has been believed for some time that the development of a cancer is a multistep process. What we have learned in recent years about oncogenes and oncoproteins places us at the threshold of understanding these steps at the molecular level.