ADVERTISEMENTS:
The following points highlight the three major types of diseases of potatoes. The diseases are: 1. Wart Disease of Potato 2. Late Blight of Potato 3. Early Blight of Potato.
Disease # 1. Wart Disease of Potato:
Wart disease of potato was for the first time discovered and described in Hungary in the year 1895. In England, the disease was first reported in 1902. It invaded the U.S.A. about 1909. In India, the disease was reported to occur in 1953 in the Darjeeling district in the variety of potato ‘Furore’ which was imported from Denmark.
The disease is essentially specific to the tubers and hardly any part above soil-level is affected. it appears on all underground parts like, buds on stems, stolon’s (Fig. 351 A), and the tubers which are the centres of infections, but never in roots.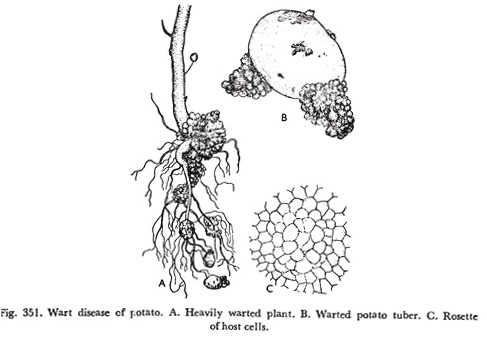
Symptoms:
Wart disease of potato is characterized by warty, tuberous, and dirty cauliflower-like proliferations of abnormal tissue of variable size and shape on the tubers (Fig. 351B) and lower parts of stems. The warty growths and contamination are spread for succeeding potato crops.
ADVERTISEMENTS:
At the infection courts there appear numerous pustules consisting of protuberances in which brownish depression centres appear.
The entry of the parasite into a particular cell causes hypertrophy and hyperplasia of the epidermal cells which are in immediate contact with that cell. Ultimately a raised ‘rosette’ overarching of host cells is formed, at the centre of which therefore the infected cell remains sunken as it itself takes no part in this outward growth (Fig. 351C).
The parasite enlarges considerably filling almost the entire cell space simultaneously resulting in the formation of wart. The centres of infection contain prosori or resting sporangia around which hyperplasia of the host tissue has been initiated. The warts are protuberances of variable size.
ADVERTISEMENTS:
A typical wart is roughly spherical, but it is usually not a solid structure. It is soft pulpy and can be cross- sectioned more readily than a tuber. Morphologically it consists of distorted, proliferated hypertrophic tissue grown together, may be partly underground and partly exposed above the soil-level.
As the warts become older, they darken somewhat and decay and the infection is spread. When affected tubers are dug, warts may disintegrate in part causing spread of infection.
Causal Organism:
This disease is induced by Synchytrium endobioticum (Schilb.) Perc., a fungus belonging to the most primitive group of fungi, the Archimycetes.
It is an obligate parasite, it starts its parasitic life as a minute uninucleate naked, uniflagellate, structure which is holocarpic, can behave as a zoospore and cause infection to the host cell resulting in the formation of wart around the infected cell. On entering the host cell, the cytoplasm of the parasite enlarges and is surrounded by a golden brown thick wall and is known as a prosorus.
The prosorus after undergoing various changes develops into a sorus of sporangia or gametangia or both. The sporangia liberate zoospores which cause fresh infection in the host cells. Whereas, the gametes that are liberated out from the gametangia after undergoing planogametic isogamy develop into zygotes which infect host cells in a process similar to that of zoospores.
A thick wall is laid down surrounding the zygotic nucleus and cytoplasm to form a resting sporangium. With maturity of the resting sporangium, the diploid nucleus undergoes meiosis and with continued mitotic division of the daughter nuclei innumerable nuclei are formed around which zoospores are organized.
The zoospores on liberation from the sporangium spread infection of the host cells. In the life history of the causal organism there are two resting stages through which it tides over unfavourble conditions and thus perennates from year to year.
The development of the two resting stages—prosori and resting sporangia of the causal organism may or may not follow each other in strict sequence. S. endobioticum, besides infecting potato, also attacks Lycopersicum esculentum, Solanum alatum and S. nigrum.
Disease Cycle:
The perennating organs—prosori and resting sporangia of the causal organism which are very resistant to extremes of temperature, high and low, may remain viable in soil even in the absence of a potato crop for many years, periods of 9 to 12 years being recorded in many instances.
ADVERTISEMENTS:
They may be carried along with manure from one place to another by any means that ensures the transport of contaminated soil and manure, adherent to seed tubers or on the underground parts like stolons being the source of infection in different localities. Even the tubers of very resistant varieties may not be free from infection.
The moderately resistant varieties in which warts are readily overlooked are most effective in spreading the pathogen.
Prosori and resting sporangia are commonly produced in small warts which cause little or no commercial loss to the potato crop but are responsible for the spread of the disease.
The disease is more prevalent during wet season than the dry since a high degree of soil moisture is necessary to bring about infection. A periodic flooding followed by drainage at a relative soil humidity of 45 per cent, and aeration are very favourable for the disease incidence.
ADVERTISEMENTS:
The entrance of the parasite takes place through a pore formed on the cell wall of the epidermal cell of the host tissue. Infection is limited to a temperature range of 12°C. to 24°C. and over a range of soil reaction from pH 3’9 to 8’5 but from neutral to slightly acid soil is most favourable.
The organism is sensitive to high degrees of alkalinity. Certain amount of moisture is essential for the germination of prosori and resting sporangia, liberation of swarmer’s and ultimately causing infection on the host cells.
With the favourable moisture conditions, temperature and soil reaction the rate of infection may be very high and widespread. The protuberances that are formed due to primary infection are the centres of secondary inoculum. Both primary as well as secondary inocula may spread infection at the same time with favourable environmental conditions inducing the disease and causing destruction of potato crop.
Disease cycle of Wart disease of potato is presented in Figure 352.
Control:
Various degrees of quarantine measures may be practised in prohibiting the export of potatoes from those localities where wart is known to occur. The disease has been successfully controlled by the introduction of potato varieties which possess complete resistance to or immunity from it.
In resistant varieties, if invasion of pathogen occurs, it dies without producing prosori and resting sporangia. A resistant variety may deteriorate or breakdown its quality when it is grown at a place which is different from where it is raised, still such a variety is more suitable than the susceptible one.
Since the resting sporangia are viable in soil even upto periods of 9 to 12 years no system of crop rotation can starve them out of the soil. Various experimental results have indicated that application of soil treatment to eradicate the disease is too expensive and impracticable to apply.
The application of sulphur has often given good results by the formation of thiosulphuric acid in the soil, so also the application of ammonium thiocyanate to the soil, but it has a retarding effect on the growth of the potato crop. But the application of lime to raise pH of the soil and the use of soil fungicides before plantings often reduce infection rate.
ADVERTISEMENTS:
Again none of the ordinarily used seed disinfectants is capable of destroying the resting sporangia in soil adherent to the tubers. Thoroughly cleaning the cultivation plots and burning the previous year’s plant debris also help to reduce the rate of infection.
Disease # 2. Late Blight of Potato:
This is one of the most serious of all the diseases of potato when conditions are favourable for its development. Since control measures are now better understood, it is possible to ward off such a dangerous disease which at one time caused severe epidemic on the European continent.
It is difficult to trace with certainty the history of late blight of potato disease. But it is probable that it originated in South America, the native home of the potato plant. There is evidence that a disease of potatoes which might have been late blight, existed in the Andes Mountains of South America as early as 1571.
It was introduced into Europe and North America sometime between 1830 and 1840. In succeeding years it increased in extent and severity, culminating in a widespread epidemic on the European continent in 1845.
The notable Irish famine of 1845 and 1846 was caused due to the destruction of the potato crop by the late blight of potato disease. It is probable that the disease was introduced into the United States and Canada at about the same time as the European introduction. This disease is now known in all potato- growing sections of the world.
In India, late blight of potato disease was first introduced in the Nilgiri hills during 1870 and 1880 and subsequently detected in Darjeeling and Hooghly.
Symptoms:
ADVERTISEMENTS:
The disease attacks the tops at about blooming time causing a blight and may also invade the tubers and cause a dry or wet rot. It damages potato crops by killing the foliage and so reducing yield, and by rotting tubers.
The symptoms on leaves, stems, and pedicels are similar in character. The first indication of the disease on leaflets, petiole, or on stem consists of brownish- to purplish-black lesions (Fig. 355A). The lesions are not delimited in size and under favourable conditions enlarge rapidly. If weather conditions are favourable, these infected areas enlarge rapidly so as often to involve the whole surface.
Extension to the stem quickly occurs in bad cases, and the entire crown- may fall over in a rotten pulp in a few days.
The influence of the weather is most marked. In dry, clear weather successful infections are limited in number and the resulting spots remain small, brown and dry, while the stems may escape altogether. In warm, humid weather the colour rapidly changes to, black, the lesions are wet, the stems are quickly attacked, and a pronounced smell of decaying vegetable matter is given off.
This characteristic odour is a helpful diagnostic character in identifying the disease. The fungus forms a whitish layer on the infected host tissue consisting of the sporangiophores bearing sporangia in great number. This growth is scanty or even absent in dry weather and dense but often evanescent on moist, cloudy days.
The diseased area turns darkar and may become dry, blackened and shrivelled if the weather becomes dry. With the return of favourable weather conditions, the fungus becomes active, and corresponding symptoms and progress reappear. When the blight is advancing rapidly in a potato field, a mildly pungent characteristic odour is given off.
ADVERTISEMENTS:
The underground parts, especially the tubers, are also affected. A brown to purple discolouration of the skin followed by a brownish dry rot extends to about 1/2 inch below the surface of the affected tubers. The dry rot does not soften the tissues but causes rusty-brown markings just below the skin and extends inwards for a variable distance in an irregular fashion (Fig. 355F).
A wet rot is usually set up by the action of secondary organisms which follow late blight development. In moist atmosphere, white tufts of mycelium and sporangiophores of the fungus appear on the surface of the infected tubers. Many of the tubers may decay before harvest. This wet rot may continue to spread in storage by contact, but the blight infection by itself does not pass from tuber to tuber.
Tuber infection is much heavier in clay soil than in sandy soil.
The losses due to late blight are of two kinds actual reduction in yield due to attacks on foliage and stem, and damage to tubers. If the leaves and stem are blighted down before the crop is mature, the tubers are prevented from reaching normal size and the yield is reduced proportionately. Sometimes great loss is sustained on account of direct attack on the tubers themselves.
Causal Organism:
The fungus was described by Montagne in 1845 as Botrytis infestans. De Bary in 1876 gave its name, Phytophthora infestans. De Bary worked on the parasitism of the organism and its causal relation to the late blight disease.
The aseptate mycelium of the pathogen ramifies in the host tissue both intra-and intercellularly. It often produces rudimentary haustoria in foliage cells (Fig. 355E). But in potato tuber cells the haustoria are more elaborate, being club-shaped, hooked or spirally twisted. The sporangiophores arise directly from the internal mycelium] emerging from the leaf through the stomata in groups (Fig. 355B to D).
Emergence through or between the cells of the epidermis is also sometimes found. On the tubers they mainly arise from lenticels or abrasions in the rind. The sporangia are colourless papillate, lemon-shaped. When the fungus is active and environment is favourable, the sporangia germinate by the development of biflagellate secondary zoospores.
Otherwise germ tubes are given out from the sporangia and thereby the sporangia behave as conidia. The zoospores when formed, after a swarming period come to rest and germinate by germ tubes which penetrate through the stomata or directly through the epidermal cells of the host tissue.
Sexual reproduction is oogamous. Oospores were first seen by Clinton in 1911 in artificial cultures of the fungus but later were obtained on diseased tubers under natural condition by Murphy in 1927 from Ireland. But Barrett (1948) has indicated the heterothallic nature of the fungus, when he was able to produce oospores by mating compatible strains of Phytophthora infestans.
Oospores are very rarely found in Europe, have recently been discovered in quantity in Mexico, where compatible strains of the fungus coexist.
Disease Cycle:
How the fungus perennates has long been one of the classic problems of plant pathology. Various theories have been advanced to explain the annual occurrence of the disease. The explanation most generally accepted has been that proposed by De Bary that the fungus could perennate in diseased tubers and then invade developing sprouts to form lesion and sporangia above ground.
The mycelium from infected seed potatoes may grow under suitable conditions to produce sporangiophores and sporangia. The sporangia so produced on coming in contact with new foliage through the agencies like wind and rain cause infection and thereby the disease is spread. Since oospores are formed in nature, there is a possibility that the pathogen perennates in that stage also.
Primary inoculum of the disease in the field usually comes from the planting of infected tubers and from oospores in the previous year’s plant debris. It has been conclusively established that the mycelium developed during previous year’s crop infection, frequently survives in the tubers without causing any great damage.
On planting the diseased tubers, the enclosed mycelium renews active growth passing into the tissues of the young sprouts chiefly between the cells of the cortex and producing sporangiophores and sporangia on the sprouts above ground if the weather conditions are favourable.
The fungus establishes itself more quickly on young than on old foliage. This tendency for the infection to start at the base of the plant is due largely to the more favourable conditions of the microclimate, such as abundance of humidity provided by the dense growth in that situation.
It has long been known that the temperature and humidity of the environment play important part in the occurrence of late blight epiphytotics. Aerial emergence of the hyphae takes place only at an atmospheric humidity exceeding 85 per cent., while sporangia apparently form only above 95 per cent, and do not become abundant under about 97 per cent.
The most favourable temperature for the production of sporangia is about 18°C. to 22°C. and for development of zoospores about 6°C to 15°C., but direct germination by germ tube occurs at an optimum of 24°C. Conditions favourable for the germination of sporangia are also optimum for host infection.
Darkness often inhibits production and germination of the sporangia. Germ tubes developed from zoospores or directly from sporangia may directly cause infection or may develop into infection hyphae.
The infection hyphae may enter the leaves or stems through the stomata or even penetrate the unbroken epidermis. In tuber infection, the entrance is probably through the lenticels. This is how the primary infection is completed. The inocula (sporangia producing germ tubes and zoospores) produced during primary infection constitute the secondary inocula which induce host infection and secondary cycles.
The sporangia are developed on the sporangiophores protruding out through the stomata of the infected host tissue. Slightly higher temperature favours growth after infection, but protracted cold weather is unfavourable. The hyphae of the intercellular mycelium send small filamentous haustoria into the host cells.
The rate of mycelial growth in the host tissues is directly proportional to the water content of the host tissues or perhaps to the ratio of water to nitrogen. There is evidence that the plants are more susceptible to the pathogen when nitrogen is withheld and growth is most rapid at 20°C. to 23°C.
The Susceptibility of the Host is Governed by Factors like:
(a) The water nitrogen ratio—increased nitrogen supply affords resistance,
(b) Water content of the leaves- succulence favours infection,
(c) Soil-moisture—dry soils increase resistance, and
(d) Supply of potash—a shortage reduces infection rate.
Diseases cycle of late blight of potato is presented in figure 356.
Control:
Late blight of potato disease can be effectively controlled by two principal methods:
(a) Direct attack on the pathogen by spraying and dusting of fungicides, and
(b) Cultivation of resistant varieties.
Following are some of the important recommended control measures of the disease:
(i) Sanitary Measures:
Previous year’s plant debris should be thoroughly cleared to cut the source of primary inoculum.
(ii) Use of Disease-Free Seeds:
Since the pathogen perennates as mycelium in the tubers, seed tubers should be used raised from disease-free fields.
(iii) Improvement of Storage of Seed Potato:
Seed tubers should be disinfected with 0-1 per cent, mercuric chloride immediately after harvest. They should be stored at 40 °F or below to check rot of tubers. The tubers should be stored in a dry-and well-ventilated place.
(iv) Soil Management:
(a) Frequent earthing-up of growing crop at a four to six inches high ridging diminishes the risk of tuber infection, (b) Spraying of soil with 10 to 20 per cent sulphuric acid or 5 per cent, copper sulphate, or 12 lbs. of copper sulphate with 1/2 lb. of caustic soda in 40 gallons of water at the rate of 100 gallons per acre cuts down infection rate.
(v) Use of Fungicides:
The best method of control of the disease incidence, is foliage spraying with suitable fungicides. In areas where late blight is common, spraying of the crop with suitable fungicides should be started well ahead of usual time of appearance of the disease. However, in order to control this disease, it is better to start spraying the crop when it is about six weeks old or when the plants are 6 to 8 inches high.
Spraying should be repeated every ten to fifteen days, depending on the weather. If there is continuous rain, the spray may be washed off. In such cases spraying may have to be repeated immediately and also done more frequently. More sprayings may be needed if the disease is in severe form.
The spray must be delivered with considerable force in the form of a fine mist so that both surfaces of the leaves and other above ground parts are completely covered by the fungicide. Bordeaux mixture has been the most and effective spray material to control the disease incidence.
The earlier spraying should be done with 4:4:50 formula of the mixture while later sprayings should be done with 6:6:50 strength. Commercial copper fungicides such as, Cupravit, Fycol 8E, Perenox, Blitox-50 (strength 1 lb. in 20 gallons of water), Fytolan (Strength 4—8 oz. in 10 gallons of water) are also used for foliar spray material.
The copper fungicides are being replaced by more effective fungicides like Dithane D-14, Dithane Z-78 (strength 15 lb. in 100 gallons of water) and Dithane M-22. Depending upon the type of spraying equipment, the quantity of liquid required for one acre will vary from 70 gallons to 300 gallons.
(vi) Improvement of Harvesting Practice:
Due precautions must be observed at harvest time to prevent tuber infection. If the foliage has been blighted, digging should be delayed until the foliage has died and dried out. Care should be taken to prevent the tubers from coming in contact with the diseased foliage. Visibly infected tubers should be sorted out from the healthy ones before storage.
(vii) Use of Resistant Varieties:
The use of resistant varieties is a very important control measure. Potato varieties in which demissum resistance has been fixed produce promising result.
Resistance Against Potato Blight:
When blight first struck the potato fields of Europe in the 1840’s, Phytophthora infestans met a host that had existed for centuries in a blight-free world.
The potato varieties were very susceptible, and great havoc was brought. Since then conscious and unconscious selection has raised the level of resistance considerably. Most of the old varieties mature late and are intensely susceptible to blight. Large-scale selection and breeding work were started to get blight resistant varieties.
It has been found that in Central Mexico Solanum demissum is prevalent at high altitudes as an uncultivated weed. Many clones of Solanum demissum exist which differ considerably in resistance to Phytophthora infestans. Potato breeders started using widely Solanum demissum as a source of R-genes for commercial varieties to be incorporated in them.
The interaction of R-genes with races of P. infestans has been proposed by W. Black and others in 1953 is now internationally accepted. The interrelationships of genes and races of P. infestans are, for example, the gene R1 makes a potato variety resistant to all races of P. infestans. that do not have the number 1 in them: races (0), (2), (3), (4), (2,3), (2,4), (3,4), and (2,3,4).
It leaves the variety, susceptible to races with the number 1 in them: (1), (1,2), (1,3), (1,4), (1,2,3), (1,2,4), (1,3,4), (1,2,3,4).
Similarly a potato variety with the three genes R1, R3, R4, is susceptible to races with the three numbers 1, 3, and 4 in them: races (1, 3, 4) and (1, 2, 3, 4), but resistant to all races without these three numbers.
This system was proposed at a time when only the four genes, R1, R2, R3, R4 had been much studied. At least two more genes, R5 and R6, are now known. The system is readily extended to include them, e.g., R1 R2 R3 R4 R5 R6 types are susceptible only to races (1, 2, 3, 4, 5, 6). The system refers only to resistance in the normal, foliage. Tuber resistance does not follow the pattern in the foliage exactly, and tubers may be susceptible to races that cannot attack the foliage.
The Mutability of Races of Infestans:
When genes for resistance from S. demissum were first incorporated in the common potato, they conferred resistance amounting almost to immunity from P. infestans for a while. In the breeder’s nurseries the new varieties with S. demissum genes behaved excellently. But as soon as the varieties had been increased to a few acres, blight began to appear conspicuously. From the blight lesions new races were isolated.
Since then it has been realized that, in conditions that favour blight, new derivatives from S. demissum invariably succumb before long to appropriate races of the blight fungus. Quite possible that the blight population inter-crossed freely in nature with sexual recombination new races are formed.
Disease # 3. Early Blight of Potato:
This is one of the most common foliage diseases of potato. The disease is designated as ‘early blight’ to distinguish it from the late blight caused by Phytophthora infestans. But it is also known as ‘leaf spot’ and ‘Alternaria blight’.
This disease was first recorded in the United States in 1891 and has since been reported from Mexico, Canada, from all the other continents and from Java, Bermuda, Australia and New Zealand. It probably occurs in all potato-growing countries of the world.
Symptoms:
The disease is characterized by the appearance of spots on the leaflets. The spots are dark-brown to almost black and are ovate or angular in shape usually 3 to 4 mm in diameter. They are circular to angular dead areas with concentric rings or ridges producing a characteristic target-board effect (Fig. 381B & C).
There is usually a narrow chlorotic zone around the spot, which fades into the normal green. The spots may be few in number and small or they may be numerous and involve a large part of the leaf area, owing to the coalescing of adjacent spots.
When spots are numerous, the general effect on the leaflet is such as to make it go into a senescent condition. Lowest leaves are affected first, and the disease progresses upward. Affected leaves may show ragged margins or irregular margins or irregular breaks, or, sometimes, perforations due to the breaking away of the dead tissue and sometimes more or less curling or rolling.
Under certain conditions, the spots may remain small without the target-board effect, being limited by some of the smaller veins. In severe cases of infection a complete blighting of the affected leaves may be resulted, preceded by the yellowing of the tissue between the spots.
On potato tubers, the lesions are slightly sunken, circular to irregular in shape (Fig: 381 A). 
Spots may develop on Stem and affected plants turn yellow and dry up. The disease may cause heavy loss to the potato crop, often even heavier than late blight. With severe foliage injury, the tuber may remain small, immature, soft-skinned, deficient in starch having poor storage quality.
Causal Organism:
Early blight is caused by Alternaria solani (Ell. and G. Martin) L. R. Jones and Grout, a fungus belonging to the Deuteromycetes.
The fungus possesses septate branched mycelium which becomes dark-coloured with age. The mycelium usually, ramifies in the intercellular spaces and also penetrates cells of the host tissue. After the invaded tissue has been filled with mycelium relatively short dark-coloured septate and erect conidiophores emerge from the stomata or arise from the older diseased host tissue.
Conidia are beaked, muriform, dark coloured, borne singly or in chains (Fig. 381D).
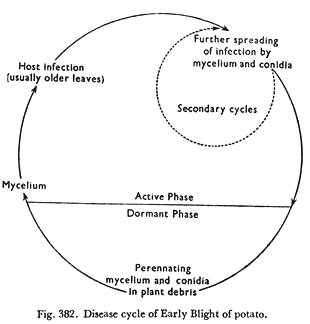
Disease Cycle:
The source of primary inoculum for the primary infection in an early crop is either the conidia which remain viable for as long as seventeen month or new conidia developed from mycelium that has persisted in the soil or in the infected leaves of the previous year’s crop. Infected potato tubers also should not be over looked as a source of inoculum.
Conidia germinate at an optimum temperature on 28°C. to 30°C. Host penetration of the fungus as a rule is through the stomata (Fig. 381D), but direct penetration through the epidermis is not infrequent. Under conditions of favourable temperature and moisture, the period of incubation is relative short, spots become visible within forty-eight to seventy-two hours after inoculation.
But conidia are produced within the next three or four days. Conidia produced during primary infection form secondary inoculum for secondary infection, and are readily detached and carried to later maturing plants by air currents. Often insects facilitate infection by producing wounds and at the same time they may also carry conidia or their bodies.
The foliage is usually most susceptible after the plants have passed their period of greatest vigour and are being weakened by external conditions or by tuber formation.
Disease cycle of Early Blight of potato is presented in Figure 382.
Control:
Infected plant debris of the previous year should be thoroughly cleaned and burnt to cut down the source of primary inoculum. Disinfection of seed is essential. It is accomplished by treatment of seeds with new improved Ceresan, applied either in 1; 2000 concentration or as a dust at the rate of 0.5 per cent, of the weight of the seed.
The use of Bordeaux mixture to some extent reduces the disease incidence. But greater success is attained with fungicides like, zinc carbamites and calcium arsenate (one pound in fifty gallons of water).
Crop rotation is an important control measure. But because of the wind-borne conidia the incidence of the disease may be delayed than completely controlled by crop rotation.