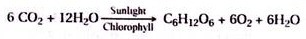ADVERTISEMENTS:
Here is your essay on Photosynthesis!
[I] Photosynthesis:
Photosynthesis is one of the most fundamental biological reactions.
The chlorophyll bearing plants trap the free energy of sunlight as photons and transform and store it as chemical potential energy by combining CO2 and water.
ADVERTISEMENTS:
The end products of photosynthesis are carbohydrates with loss of oxygen. These directly or indirectly serve as the source of energy for all living beings, except chemosynthetic bacteria.
Photosynthesis as an oxidation-reduction reaction:
In 1931, С. B. Niel suggested that water is the hydrogen donor in the oxidation-reduction that occurs in photosynthesis. The ratio of oxygen evolved to carbon dioxide consumed is one. The over all reaction of photosynthesis is —
nН2О + nСО2 → light / chlorophyll nO2+ (CH2O)n
ADVERTISEMENTS:
Thus, water is the donor of H2. The O2 that is liberated also comes from water and not from CO2 as has been confirmed by experiments using 18 ‘heavy oxygen – 18O’.
Thus, in this reaction, CO2 acts as an electron acceptor (A) and H2O as an electron donor (D). The energy contained in the visible light portion between 400 and 700 nm is transmitted in discrete particles called photons. A photon contains one quantum of light energy. Photons with shorter wave lengths have higher energy. The quantasomes are thought to be the units which absorb light quanta and contain chlorophyll in association with lipid.
Photosynthesis involves a complex series of reactions, some of which take place only in the presence of light, while others can also be carried out in the dark.
(1) Light or photochemical reactions.
(2) Dark reactions.
In 1937, R. Hill (British Botanist) was the first to provide evidence that the photochemical reactions of plant cells took place in chloroplasts. Later it was discovered that the oxidized form of the coenzyme NADP+ was the normal hydrogen acceptor of the photochemical reaction, producing NADPH.
Arnon (1954) showed that in the presence of light, chloroplasts could make ATP from ADP and Pi (this is the process called photophosphorylation). In photosynthesis, the electrons flow from H2O to NADPH. The photochemical reaction takes place in the thylakoid membranes.
1. Light or photochemical reactions:
Robert Hill found that when isolated chloroplasts illuminated, they produced oxygen and acquired reducing properties. Carbon dioxide was not involved and water had been split into hydrogen and oxygen.
This is now called the Hill reaction. It is the main event in light reactions of photosynthesis.
The function of light reactions is two fold —
(1) The photochemical splitting of water provides hydrogen atoms for the reduction of CO2, and
(2) Producing of ATP which provides energy for the subsequent synthesis of carbohydrates.
ADVERTISEMENTS:
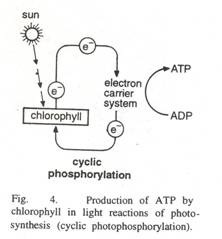
When light strikes the chlorophyll molecule, the energy level of an electron is raised. In this ‘excited’ state the electron is emitted and taken up by an electron acceptor. The electron is then passed through a series of carriers, the last one handing it back to the chlorophyll molecule.
In losing an electron the chlorophyll becomes positively charged and unstable, but neutrality and stability are restored when the electron is returned. Thus the electrons emitted from chlorophyll are returned to it unchanged and energy being released as heat and fluorescence.
This cyclic process is known as cyclic photophosphorylation. It is concerned with synthesizing ATP molecules but the source of energy is different. In photophosphorylation energy comes from sunlight via chlorophyll.
Splitting of water:
ADVERTISEMENTS:
We have already seen that when light strikes chlorophyll an electron is emitted. Now not all the electrons are returned via the electron carrier system to chlorophyll. Sometimes an electron, together with a hydrogen ion, is taken up by a hydrogen acceptor, nicotinamide adenine dinucleotide phosphate (NADP), which is thus reduced.
The reduced NADP (i.e., NADPH2) now enters the dark reactions of photosynthesis, handing on the hydrogen which is then used in the reduction of CO2. This hydrogen comes from the dissociation of water into hydrogen (H+) and hydroxyl (OH–) ions in light reactions.
When NADPH2 loses its hydrogen, the reduced NADP becomes reoxidised and can be used again.
ADVERTISEMENTS:
The chlorophyll molecule which has lost an electron is in an unstable state. The hydroxyl ion donates an electron to chlorophyll and the OH resulting from this forms water and oxygen.
OH– + chlorophyll+ chlorophyll + OH
4(OH) → 2H2O + O2
By acting as an electron donor the hydroxyl ion restores the stability of the chlorophyll molecule. The oxygen is given off in photosynthesis.
The electron derived from the hydroxyl ion is conveyed from one chlorophyll molecule to another via an electron carrier system. This results in the formation of ATP. This pathway is non-cyclic phosphorylation.
The passage of electrons in non-cyclic phosphorylation is complex, involving two distinct molecules (or groups of molecules) of chlorophyll a. These are known as photosystems I and II. They are at different energy levels, and under the influence of light the energy state of an electron can be raised, enabling it to be transferred from one system to the other. In the course of its journey it passes through electron, carriers with resulting ATP synthesis. One of the electron carriers is an iron-containing protein called ferredoxin.
ADVERTISEMENTS:
The various pigments [Chlorophyll a (blue-green), chlorophyll b (yellow-green), xanthophyll (yellow), carotene (orange) and phaeophytin (grey) is the breakdown product of chlorophyll.] act as electron carriers. It appears that electrons are moved from one pigment to another. Chlorophyll a is the final pigment in the series, receiving electrons from other pigments and handing them on via electron carriers to NADP.
In the Figure 5, photosystem I (groups of molecules of chlorophyll a) is at a higher energy level than photosystem II. When light strikes a chlorophyll molecule belonging to photosysiem I, an electron is raised to a higher energy state and is taken up by an electron acceptor and then passed to NADP.
Meanwhile the electron which has been lost from photosystcm I is replaced by an electron from photosystem II. This later electron, lost from photosystem II under the influence of light is taken up by an electron acceptor and then passed to photosystem I via the electron carrier system with the production of ATP. The electron lost from photosystem II is replaced by an electron from the hydroxyl ion derived from the splitting of water.
Dark reactions (carbon-reduction cycle):
Dark reactions involve the reduction of carbon dioxide to form carbohydrate. This is an endergonic process requiring energy. The energy is supplied by the hydrolysis of ATP formed in the light reactions and also from NADPH2 which now carries high energy electrons. The NADPH2 also provides the hydrogen for reducing the carbon dioxide.
The reduction of CO2, and subsequent synthesis of carbohydrate, takes place in a series of small steps, each controlled by a specific enzyme. The individual steps have been analysed by Melvin Calvin and his associates.
ADVERTISEMENTS:
The chain of reactions is cyclical and known as the Calvin cycle. In the first step the CO2 combines with a 5-carbon organic compound called ribulose biphosphate (RUBP). This is carbon dioxide acceptor and fixes the CO2, i. e., incorporates it into the photosynthetic machinery of the plant.
The enzyme needed for this is called RUBP carboxylase. The combination of carbon dioxide with ribulose biphosphate gives an unstable 6-carbon compound which splits immediately into two molecules of a 3-carbon Compound, phosphoglyceric acid (PGA). Later the PGA is reduced to form a 3-carbon sugar, phosphoglyceraldehyde (PGAL).
The hydrogen for the reduction comes from reduced NADP of light reaction, which also supplies most of the energy, the rest coming from ATP. The 3-carbon sugar is now built up to a 6-carbon sugar which can be converted into starch for storage.
Not all the 3-carbon sugar (PGAL) is converted into 6-carbon sugar. Some of it (majority infact) enters a series of reactions which results in the regeneration of ribulose biphosphate. Its continuous supply is important for the continued fixation of CO2.
The energy balance of photosynthesis is:
6CO2+ 12H2O → light, chlorophyll C6H12O6+ 6O2+ 6H2O
It represents a storage of 686,000 calories per mole. This energy is provided by a total of 12 NADPH and 18 ATP molecules, representing 750 Kcal.
The starch is not the only end-product of photosynthesis. In Calvin’s experiment radioactive carbon was eventually identified in other compounds including amino acids. For amino acids formation, nitrates are required.
These are converted to ammonium ions which are used for the formation of glutamine. Fnm this other amino acids are made by transamination. In transaminat on, the amino group is removed from amino acid and transferred to a carbohydrate derivative which is thereby converted into a new amino acid.
C4 plants:
Some plants like cane-type plants such as sugar cane and maize, use another compound, phosphoenol pyruvic acid (PEP) instead of ribulose biphosphate, as the substrate for carbon dioxide fixation in certain of their cells. The immediate product of CO2 fixation is not 3-carbon PGA but the 4-carbon compound oxaloacetic acid. Because of this, such plants are called C4 plants in contrast to those that produce 3-carbon PGA which are known as C3 plants.
The oxaloacetic acid of C4 plants is subsequently converted into malic acid, from which CO2 is fed into the Calvin cycle to form carbohydrate.
Meanwhile, so long as light is available, the PEP can be resynthesized (Hall Hatch and Roger Slack).
(a) Light reaction:
(i) Photolsis: H2O → light 2H+ O
NADP or TPN + 2H→ NADPH or TPNH + H+
(ii) Phosphorylation: 2ADP + 2 phosphate → 2 ATp
(b) Dark reaction:
(iii) Formation of phosphoglyceric acid (PGA):
ATP + Ribulose pentosephosphate → phosphopentokinase RDP + ADP (ribulose diphosphate)
Pentose diphosphate + CO2+ H2O
Glucose → isomerase, 2 PGA
[II] Food storage:
Some unpigmented plastids like leucoplasts store the essential food materials like protein, oil and starch. Later on these are used during germination of seeds and development.
[III] Hereditary carrier:
Recent studies show that these plastids, like chromosomes, are transmitted directly to the daughter cells during cell division. Cytoplasmic inheritance of plastids in Mirabilis is the well-known example. They produce phenotypic effects in Oenothera and other plants.
[IV] Chloroplasts as semiautonomous organoid:
The chloroplast matrix contains dissolved salts and enzymes of photosynthesis. Besides these, like mitochondria, it contains RNA, DNA and ribosomes, and is capable of carrying on protein synthesis.
The chloroplast ribosomes are of the same size as ribosomes in prokaryotes. Chloroplasts are also semi-autonomous like mitochondria. They can grow and divide, and their DNA contains a portion of the genetic information needed for the synthesis of chloroplast proteins.
[V] Inheritance of chloroplasts:
Cells have the capacity to outgrow their chloroplasts and the rate of multiplication of chloroplasts is partly independent of the rate of multiplication of entire cells. Brawerman and Chargaff (1960) discovered it in Euglena gracilis after a temperature shock.
Cells which were permitted to multiply rapidly became irreversibly bleached, whereas cells prevented from dividing regained their normal ability to produce chloroplasts. They concluded that Euglena contains an autonomously replicating factor which is necessary for chloroplast formation.
[VI] DNA in chloroplasts:
Chloroplasts contain both DNA and the necessary mechanism for synthesizing specific RNA’s and proteins from a DNA template. DNA is found in chloroplasts (Stocking and Gifford, 1959). Ris and Plaut (1962) have also found DNA in the chloroplasts of alga Chlamydomonas. It has now been generally accepted that characteristic chloroplast DNA’s or chloroplast chromosomes occur in the photosynthetic organelles of algae and higher plants.
According to Brawerman (1966) this DNA differs from nuclear DNA in GC (guanosine and cytosine) content. Chloroplasts also contain a DNA-dependent RNA polymerase; it appears that specific RNA’s are synthesized from chloroplast DNA as a template (Kirk 1966). Chloroplasts DNA are capable of self-duplication.
[VII] Chloroplast ribosomes:
Lyttleton (1962) isolated chloroplast ribosomes, which are estimated to make up 3 to 7% of the chloroplast dry mass. Chloroplast ribosomes are smaller than cytoplasmic ribosomes. These are 60-66S. Chloroplast ribosomes also dissociate reversibly into 50S and 35S subunits, in a way that is found in E. coli ribosomes (Boardman et al., 1966). Chloroplasts have three types of RNA required for protein synthesis: ribosomal, transfer and messenger. Chloroplast ribosomes associate to form polysomes for synthesis of proteins (Gunning and Steer, 1975).
[VIII] Protein synthesis:
Protein synthesis in mitochondria and chloroplasts is similar to that of prokaryotes. For example, the size of chloroplast ribosomes is the same as ribosomes of blue-green algae, and ribosomes of chloroplasts and mitochondria more closely resemble prokaryotic ribosomes in antibiotic sensitivity than they do eukaryotic ribosomes.