ADVERTISEMENTS:
In this article we will discuss about:- 1. Introduction to Puccinia Graminis 2. Vegetative Body of Puccinia Graminis 3. Different Type of Spores Found 4. Life History.
Introduction to Puccinia Graminis:
Puccinia graminis causes stem rust of different cereals like wheat, rye, oat etc. The isolate of wheat cannot infect oat or any other host and vice versa, which indicate their host specificity. The phenomenon where a specific pathogen infects only a specific host is called biological specialisation.
Depending on the above phenomenon, the species graminis is again divided into different biological strains or biological forms or subspecies.
ADVERTISEMENTS:
Later, Stakman and many others designated the biological strains or subspecies as formae specialis abbreviated as f. sp, as named by trinomials i.e., the addition of third name with binomials after considering the chief host of the pathogen. So the name Puccinia graminis which infects wheat (Triticum aestivum) is called as Puccinia graminis f. sp tritici.
Thus P. graminis is a composite species. Based on pathogenecity, size of spores and cultural variability, it has been divided into different subspecies:
Depending on the nature of parasitism on different agronomic varieties of the host, the subspecies is again divided into physiological races. All the above biological strains/forms and physiological races have the common barberry plant as their alternate i.e., secondary host. The differences they show are the graminaceous host.
ADVERTISEMENTS:
Wheat crops of Northern and Southern region of India are largely affected by Puccinia graminis tritici. In South India (Nilgiri ranges) the disease appears in November, thereby the vegetative growth of the crop is affected mlich more than North India, where the disease appears in late March or thereafter when the crop is near its maturity. The rate of photosynthesis reduces greatly, thereby causing much loss in yield.
Vegetative Body of Puccinia Graminis:
The vegetative body is mycelium. Mycelia are of two types: dikaryotic and monokaryotic. Both the types are septate, much branched, grow intercellularly and produce special haustoria, which penetrate the host cell.
Simple pore if present in the septum, maintains protoplasmic continuity between neighbouring cells. The cell wall is composed of chitin and glucan. The cytoplasm also contains vacuoles, oil globules, glycogen bodies etc.
The dikaryotic mycelium (n + n) occurs in wheat plant i.e., the primary host and the monokaryotic mycelium occurs on barberry plant i.e., the alternate host of the pathogen.
Different Type of Spores Found in Puccinia Graminis:
Puccinia graminis tritici produces five different types of spores in its life cycle. These are uredospore, teleutospore, basidiospore, pycniopore and aeciospore.
Uredospores and teleutospores grow on wheat, the primary host; basidiospores on soil or on dead plants upon soil that developed from teleutospore; and the pycniospores and aeciospores on barberry plant, the alternate host of the pathogen.
1. Uredospore:
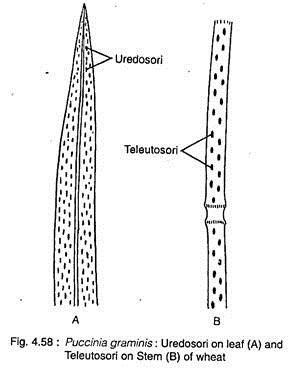
The uredospores are borne on uredosorus (Fig. 4.58A, 4.59A). The uredosorus develops on wheat plant from the dikaryotic mycelium produced by germination of aeciospore, which comes from barberry plant generally through wind dissemination. The aeciospore after reaching the wheat plant may attack leaf, stem or glumes.
ADVERTISEMENTS:
On germination, the aeciospore produces dikaryotic mycelium which enters through stomata and grows intercellularly. The mycelium of the subepidermal region develops erect hyphae, those produce binucleate uredospores at their tips.
The uredospores develop in groups under the epidermis, called uredosorus, which appear in the form of reddish-brown pustules. With maturity, the host epidermal wall bursts by pressure of developed spore and uredospores become exposed.
Uredospores are stalked, oval, unicellular, brown, thick walled with 4-round equatorial germ pores measuring outer layer 25-30 µm 17-20 pm. The spore wall is thick with echinulate outer layer (Fig. 4.59B).
The uredospores in favourable condition (i.e., in winter season) again germinate (Fig. 4.59C), thus infect the wheat plant (Fig. 4.59D) and develop next crop of uredospore. The procedure may repeat several times in a single season.
ADVERTISEMENTS:
2. Teleutospore:
At the end of wheat season, the uredosori also produce teleutospore along with uredospore. At that time the sorus is known as mixed sorus. The mixed sorus gradually converts solely into teleutosorus (Fig. 4.58B, 4.60A). Teleutosorus develops exclusively by the infection of uredospore. The teleutosori look black raised streak that developed on leaf sheath and also on stem.
The teleutospores (Fig. 4.60B) are stalked, spindle-shaped, thick and smooth-walled with round or pointed apex, 2-celled and slightly constricted at the septum. Spores are chestnut brown in colour and measure about 15-20 µm x 40-46 µm.
Each cell is dikaryotic (n + n) having one germ pore. The germ pore is at the top of the apical cell and it is below the septum in the lower cell. Both the nuclei in a cell fuse together and form a diploid (2n) nucleus.
ADVERTISEMENTS:
The teleutospores become exposed by rupturing the host epidermis. It acts as a resting spore and survives during unfavourable condition. With the onset of favourable condition of low temperature with high humidity, the teleutospores germinate on soil either detached or attached condition with the dead host.
3. Basidiospore:
It develops on germination of teleutospore. The teleutospore germinates by producing one germ tube from each cell. The germ tube has limited growth and is called promycelium, probasidium or epibasidium. The diploid nucleus passes inside the probasidium and undergoes meiosis to form four haploid nuclei, two (+) and two (-) type.
The probasidium now becomes four celled by transverse wall. Each cell then develops small projection, the sterigma through which nucleus and cytoplasm push outside and develop basidiospores, of which two are (+) and two of (-) type (Fig. 4.60C).
ADVERTISEMENTS:
Basidiospores are unicellular, thin-walled and very small. After discharge from basidium by explosive mechanism, they are disseminated by air current. The basidiospores can survive for a few days. They can infect only the leaves of alternate host, the barberry, otherwise they die.
ADVERTISEMENTS:
4. Pycniospore:
During favourable condition, the basidiospore germinates on contact with barberry leaflet towards the upper surface by producing germ tube. The germ tube penetrates the epidermis and grows there intercellularly.
The nature of the mycelium depends on the nature of basidiospore, either of + or – type. Within few days, the growing mycelium becomes aggregated under the epidermis and forms a yellowish flask-shaped structure, called Pycnium or Spermogonium (Fig. 4.61 A, 4.62A).
The Pycnium has small sterile mycelium at the neck, called periphyses, which intermingle with much larger thin-walled simple and branched receptive or flexuous hyphae.
ADVERTISEMENTS:
The bottom of the inner side of pycnium is lined by many uninucleate tapering cells, the pycmophores or spermatiophores, which develop many small oval to spherical uninucleate cells, called pycniospores (spermatia).
Depending on the nature of basidiospore, the pycnium and pycniospore may be of + or – type. Numerous pycnia of different types (+ and -) can grow in cluster on the upper surface of the leaf. The pycniospore or spermatium does not infect any host.
The mature pycnium (+ and – type) secretes nector drops during release of mature pycniospores which get intermixed.
The insects get attracted by nector and help in the transfer of pycniospore or spermatium to the flexous hyphae of opposite mating type (Fig. 4.62B). The wall at the point of contact dissolves and the nucleus of pycniospore (spermatium) passes to the flexuous hyphae, thus dikaryotic condition is established. This process is known as spermatisation.
5. Aeciospore:
During development of Pycnia, some hyphae of + and – type goes downward towards the lower side of the leaf and forms respective immature aecia (Protoaecium or aecial primodium). Further growth is initiated within few days of spermatisation.
The spermatial nuclei pass downward through the hyphae and after crossing the inner tissue they reach and dikaryotise the basal cells of the aecial primordium. The aecial primordium then stimulates to further growth and within few days, on the lower surface of the leaf, aecium (PI. aecia) or cluster cup (n + n) is formed.
The young aecium is buried inside the tissue below the lower epidermis, but with maturity it pushes and ruptures the epidermis, thereby spores are exposed. The aecium is inverted cup-shaped structure (Fig. 4.61 B, 4.62A) with outer margin composed of short cells, called peridium.
The stalk cell after becomes dikaryotised, divided mitotically into chain of alternately arranged large and small cells (Fig. 4.62A, C). The large cells form the aeciospores (n + n) and the small one becomes sterile, called disjunctor cell. The disjunctor cell helps in spore dispersal.
The aeciospores are unicellular, binucleate (n + n), thin-walled and orange in colour. The young aeciospores are polyhedral in shape, but becomes globose with maturity. They are dispersed by air current and can infect only the graminaceous host i.e., wheat plant. After falling on wheat plant (stem and/or leaf), they germinate by producing germ tubes (n + n), that penetrate inside the host tissue and establish infection.
The graphic life cycle of Puccinia graminis tritici’s presented in Fig. 4.63.
.
Life History of Puccinia Graminis Tritici:
Puccinia graminis tritici is a heteroecious, macrocyclic and polymorphic pathogen:
(a) Heteroecious i.e., it requires two unrelated hosts such as wheat (Triticum aestivum), the primary host and Barberry (Berberis vulgaris), the secondary host; to complete the life cycle.
(b) Macrocyclic i.e., it has extended life cycle, consists of five types of spores i.e., uredospores, teleutospores, basidiospores, pyncniospores or spermatia and aeciospores. [Demicyclic rust: All but uredinial stage is absent and Microcyclic rust. The teleutospore is the only binucleate spore produced. Although three forms are present, but pycnia or spermogonia may occasionally be absent.]
(c) Polymorphic (Poly, many; morph, form or structure) i.e., it produces 5 different types of spores in its life cycle.